18 December 2021: Clinical Research
Age-Stratified Cut-Off Values for Serum Levels of N-Terminal ProB-Type Natriuretic Peptide and Mortality from Sepsis in Children Under Age 18 Years: A Retrospective Study from a Single Center
Yan-na Sun1ABCE, Chun-wang Lin1ABCDEF*, Wen Tang2BF, Jiang-ping Zhang1CFDOI: 10.12659/MSM.933400
Med Sci Monit 2021; 27:e933400
Abstract
BACKGROUND: The N-terminal fragment of proB-type natriuretic peptide (NT-proBNP) is an established predictive marker for sepsis-related mortality in adult. This retrospective study aimed to determine age-stratified cut-off values for serum levels of NT-proBNP and mortality from sepsis in children under 18 years.
MATERIAL AND METHODS: Patients were stratified by age as follows: <1 year, 1-3 years, 4-6 years, and 7-18 years (age groups). The control group consisted of age- and sex-matched healthy children. Serum NT-proBNP levels were detected by laboratory assays in the participants. The appropriate serum NT-proBNP cut-off values for predicting short-term mortality of the sepsis patients were calculated via receiver operating characteristic (ROC) curve analyses.
RESULTS: Among 327 pediatric patients with sepsis, the serum NT-proBNP cut-off concentrations for predicting sepsis-related mortality in the <1 year, 1-3 years, 4-6 years, and 7-18 years age groups were 5000 ng/L, 4500 ng/L, 4100 ng/L, and 3800 ng/L, respectively (P<0.001). The area under the ROC curve (AUC) values for these were 0.815, 0.812, 0.806 and 0.725, respectively (P<0.001).
CONCLUSIONS: This retrospective study provided the age range-specific serum NT-proBNP cut- off concentrations for predicting short-term mortality in children. In children <1 year, 1-3 years, 4-6 years, and 7-18 years, age-stratified cut-off values that predicted sepsis-associated mortality were 5000 ng/L, 4500 ng/L, 4100 ng/L, and 3800 ng/L, respectively.
Keywords: children, N-terminal pro-BNP, Age-Stratification, Sepsis, septic shock, Prognostic Biomarker, Adolescent, Age Factors, Child, Child, Preschool, Female, Humans, Infant, Infant, Newborn, Male, Natriuretic Peptide, Brain, Peptide Fragments, ROC Curve
Background
The N-terminal fragment of proB-type natriuretic peptide (NT-proBNP) is released in response to increased ventricular pressure and then converted into active BNP [1]. The serum NT-proBNP concentration was first identified to be a marker for cardiac dysfunction [1–4], and later research demonstrated that the serum NT-proBNP concentration is increased in sepsis patients and can be used to predict mortality among adult patients with sepsis [5–8]. The well-established mortality rate among sepsis patients is 34.2% [9]. In studies performed in the United States and Europe, patients with severe sepsis/septic shock was determined NT-proBNP cut-offs of 4000 pg/mL [sensitivity, 0.728 (95% CI were 0.703–0.753); specificity, 0.789 (95% CI were 0.710–0.867); AUC, 0.787 (95% CI were 0.766–0.809)] optimally predicted short-term mortality in adult patients with sepsis [9]. This cut-off value has been accepted and cited in several reports [10–14]. However, the serum NT-proBNP concentration closely correlates with age, especially in children [15]. Appropriate age-specific cut-off values for serum NT-proBNP concentration for identifying heart failure in children have been determined (580 ng/l, 529 ng/l, 500 ng/l and 455 ng/l, respectively, for less than 1 year old, 1–3 year olds, 4–6 year olds and 7–18 year olds in patients) [15]. Robert d. Ross believed that serum NT-proBNP concentration greater than 300pg/ml was the starting point for heart failure score in children [16]. However, such age-specific cut-offs for predicting sepsis-related mortality in children of different ages less than 18 years have not been reported.
Therefore, the present retrospective study was conducted, in which patients were stratified by age as follows: <1 year, 1–3 years, 4–6 years, and 7–18 years (age groups); serum NT-proBNP levels were detected by laboratory assays in the participants; the appropriate serum NT-proBNP cut-off values for predicting short-term mortality of the sepsis patients were calculated via receiver operating characteristic (ROC) curve analyses. The study was used to determine appropriate serum NT-proBNP cut-off concentrations for short-term (within 28 days) mortality prediction in pediatric sepsis patients stratified by age. The calculated cut-off values can be applied to improve the management of pediatric cases of sepsis. Therefore, this retrospective study from a single center aimed to determine age-stratified cut-off values for serum NT-proBNP levels and mortality from sepsis in children under 18 years.
Material and Methods
STUDY GROUPS:
All included pediatric patients with sepsis were seen between June 2012 and December 2020 at Shunde Women’s and Children’s Hospital of Guangdong Medical University and the First Affiliated Hospital of Sun Yat-sen University. Sepsis was diagnosed based on the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3): Sepsis results in life-threatening organ dysfunction due to loss of regulation of normal organ function following generalized infection. Septic shock is identified clinically by a vasopressor response required to maintain the mean arterial pressure of 65 mm Hg or more and a serum lactate level of more than 2 mmol/L (>18 mg/dL), without hypovolemia [17]. The measured serum NT-proBNP concentration did not affect the diagnosis of sepsis in any case.
A total of 327 children (18 years of age or younger) with sepsis were included in the present study. Patients with complex congenital heart disease, nephritis, nephropathy syndrome, Kawasaki disease, pulmonary arterial hypertension disease, or genetic cardiomyopathy were excluded, because these diseases can influence serum NT-proBNP levels. The control group consisted of 115 healthy children.
LABORATORY ASSAYS:
The pediatric sepsis patients had a blood sample taken on hospital admission. Blood samples were collected from healthy children during routine check-ups. For all samples, 2 ml of venous blood was collected into a tube containing EDTA-K2, and the serum NT-proBNP concentration was measured in a blinded manner.
Serum NT-proBNP levels were measured using a fluorescence quantitative analyzer (No. 1794-2008, Response Biomedical Corp, Vancouver, Canada). Serum procalcitonin (PCT) and C reactive protein (CRP) levels were measured with a biochemical analyzer (No. p800, Roche Diagnostics, Basel, Switzerland).
STATISTICAL ANALYSES:
SPSS version 20.0 statistical software (IBM, Armonk, NY, USA) was used for all statistical analyses. The distribution of serum NT-proBNP levels was tested for normality with the use of the Kolmogorov-Smirnov (K-S) method. If the results were normally distributed, they are presented as mean±SD. If the results were not-normally distributed, they are presented as the median (M). The Wilcoxon nonparametric test was used to compare the serum NT-proBNP levels in patients with sepsis and septic shock between 2 groups, the Kruskal-Wallis H test was used compare the serum NT-proBNP levels in patients with sepsis and septic shock between more than 2 groups. Spearman correlation analysis was performed to determine correlations between serum NT-proBNP and procalcitonin levels in all sepsis patients. The appropriate cut-off values of serum NT-proBNP for predicting sepsis-related mortality in the different age groups were determined by receiver operating characteristic (ROC) curve analysis. The area under the ROC curve (AUC), 95% confidence interval (CI), sensitivity, specificity, and positive likelihood ratio values were analyzed. Significance was established at the level of
Results
CLINICAL CHARACTERISTICS OF THE STUDY PARTICIPANTS:
The 327 pediatric patients with sepsis included 171 males and 156 females with a median age of 3.2 years (range, 0–18 years). There were no significant differences in age and sex between the children in the sepsis and control groups (Z=−1.31,
The 115 healthy children in the control group included 59 males and 56 females with a median age of 3.6 years (range, 0–18 years). There were no significant differences in age and sex between the children in the sepsis and control groups (Z=−1.25,
All participants in both the sepsis and control groups were stratified into 4 age groups based on the age ranges of <1 year, 1–3 years, 4–6 years, and 7–18 years. The demographic and clinical characteristics of the children in the 2 groups are presented according to age group in Table 1.
Among the 327 pediatric patients in the sepsis group, 232 (70.90%) and 95 (29.10%) were diagnosed sepsis and septic shock, respectively. The primary diseases among the sepsis patients were serum pneumonia (n=127), meningoencephalitis (n=48), viral myocarditis (n=40), severe
VARIATION IN NT-PROBNP LEVELS IN PEDIATRIC SEPSIS PATIENTS:
K-S testing was used to analyzed the distribution of serum NT-proBNP levels. The serum NT-proBNP values were non-normally distributed in both the sepsis groups and healthy control groups (
Among all 327 patients in the sepsis group, the median serum NT-proBNP concentration measured upon admission to the hospital was 4456.7 ng/L (range, 3489.5–12 500.6 ng/L, and the median serum NT-proBNP concentrations for patients <1 year, 1–3 years, 4–6 years, and 7–18 years of age were 5348.7 ng/L (range, 5207.2–12500.6 ng/L), 4486.8 ng/L (range, 4817.9–9000 ng/L), 4107.4 ng/L (range, 4588.3–6800.4 ng/L), and 3829.6 ng/L (range, 3489.5–6047.7 ng/L), respectively. The median serum NT-proBNP concentrations differed significantly among the age groups (χ2=40.36, P<0.001, Tables 1, 2). The patients with a sepsis diagnosis other than septic shock (n=232) and patients diagnosed with septic shock (n=95) had significantly different serum NT-proBNP levels (χ2=36.17, P=0.000; χ2=38.55, P=0.000; respectively, Table 2).
Among all 115 healthy children, the median serum NT-proBNP concentration was 175.7 ng/L (range, 14.4–1286.4 ng/L), and the median serum NT-proBNP concentrations for patients <1 year, 1–3 years, 4–6 years, and 7–18 years of age were 389.8 ng/L (range, 79.3–1286.6 ng/L), 152.9 ng/L (range, 45.2–274.7 ng/L), 114.5 ng/L (range, 28.6–249.1 ng/L), and 76.5 ng/L (range, 14.4–173.9 ng/L), respectively. In the control group, the median serum NT-proBNP level was negatively correlated with age (r=−0.816, P=0.000; Figure 1).
Among the 327 patients, 228 (69.7%) patients recovered (mean length of hospital stay, 13.5 days) and 99 (30.3%) patients died (mean length of hospital stay, 6.5 days). The numbers of patients who died in the ages <1 year, 1–3 years, 4–6 years, and 7–18 years groups were 28, 40, 16, and 15, respectively. The median serum NT-proBNP levels among the recovered and deceased patients were 4223.5 ng/L (range, 3856.4–8488.3 ng/L) and 5082.8 ng/L (4435.2–12500.6 ng/L), respectively, and a significant difference was detected between these values (Z=−3.735, P<0.001, Table 2). The serum NT-proBNP level was negatively correlated with age in the 99 deceased patients (r=−0.325, P=0.000; Table 2, Figure 2). In 228 patients who recovered, the median serum NT-proBNP levels before and after treatment were 4223.5 ng/L (range, 3856.4–8488.3 ng/L) and 334.5 ng/L (range, 145.2–403.7), respectively, and the difference between them was significant (Z=−8.465, P<0.001, Table 2).
In the 95 patients diagnosed with septic shock, more fluids were administered during the first week of treatment, with patients with a sepsis diagnosis other than septic shock receiving <7000 mL, and patients with septic shock diagnosis receiving >9000 mL. In the first week of treatment, the median NT-proBNP levels among all patients with a sepsis diagnosis other than septic shock and those with a diagnosis of septic shock were 3947 ng/L and 5835 ng/L, respectively (
The serum procalcitonin levels measured during hospital admission were 15±1.8 mg/L, 16±1.4 mg/L, 14±0.7 mg/L, and 13±0.9 mg/L in the sepsis patients aged <1 year, 1–3 years, 4–6 years, and 7–18 years, respectively. Moreover, an increase in the serum NT-proBNP level was observed with an increasing serum procalcitonin level, and a positive correlation was found between these factors for all age groups (r=0.586, r=0.551, r=0.497 and r=0.477, all P<0.001; Table 3).
AGE-SPECIFIC SERUM NT-PROBNP CUT-OFFS FOR PREDICTING MORTALITY IN CHILDREN WITH SEPSIS:
The ROC curves for the ability of serum NT-proBNP to predict 28-day mortality in pediatric sepsis patients of differing ages are presented in Figure 3. The AUC with 95% CI values were 0.815(0.73–0.93), 812(0.69–0.92), 0.806(0.66–0.91) and 0.725(0.62–0.91), respectively for ages <1 year, 1–3 years, 4–6 years, and 7–18 years. The sensitivity and specificity of serum NT-proBNP were 0.786 and 0.937, 0.807 and 0.915, 0.814 and 0.882, 0.847 and 0.837, respectively for ages <1 year, 1–3 years, 4–6 years, and 7–18 years. The optimal cut-off values of serum NT-proBNP levels were 5000 ng/L, 4500 ng/L, 4100 ng/L, and 3800 ng/L for ages <1 year, 1–3 years, 4–6 years, and 7–18 years, and the difference between them was significant (χ2=38.12, P<0.001). The positive likelihood for the optimal cut-off values were 21.6, 17.3, 20.2 and 19.7 for ages <1 year, 1–3 years, 4–6 years, and 7–18 years. The AUC with 95% CI values, sensitivity and specificity values, optimal cut-off values, and positive likelihood ratios calculated from the ROC curve analyses are presented in Table 4.
The AUC values tended to decrease with increasing age, whereas the sensitivity tended to increase with age. Conversely, the specificity of NT-proBNP levels for the prediction of sepsis-related mortality tended to decrease with age. Likewise, the positive likelihood ratio decreased with age. The most important findings were that the optimal cut-off values for the use of serum NT-proBNP to predict sepsis-related mortality varied among the age groups. The age range-specific serum NT-proBNP levels were 5000 ng/L, 4500 ng/L, 4100 ng/L, and 3800 ng/L for predicting short-term mortality in pediatric sepsis patients <1 year, 1–3 years, 4–6 years, and 7–18 years of age, respectively, and these values for the different age groups differed significantly.
Discussion
The age-range-specific optimal serum NT-proBNP cut-offs for detecting heart failure in adults were previously determined to be 450 pg/mL for patients <50 years, 900 pg/mL for patients 50–75 years, and 1800 pg/mL for patients >75 years. For children, these reported cut-off values were 580 ng/L for ages <1 year, 529 ng/L for ages <1–3 years, 500 ng/L for ages 4–6 years, and 455 ng/L for ages 7–18 years [15,16,18]. Interestingly, these results indicate that the appropriate NT-proBNP cut-off value for identifying heart failure in patients over 50 years old increases with age, whereas that in children under 18 years of age decreases with age.
Age has a critical influence on serum NT-proBNP levels. Previous research showed that serum NT-proBNP levels were highest during the first few days after birth in babies, then gradually decreased throughout infancy and childhood, and similar findings were found in patients with heart failure [16,19]. These findings suggest that the predictive value of plasma NT-proBNP levels varies with age, and age-stratification must be performed to determine the diagnostic cut-off values.
Substantial research has shown that serum NT-proBNP level is an independent predictor of mortality in patients with sepsis [5,7,10,13,14,20], and the established cut-off value for adult patients is 4000 pg/mL [9–14]. However, age-specific NT-proBNP cut-off values, as reported for the detection of heart failure [15–18,21], have yet to be reported for prediction of sepsis-related mortality in children.
The effect of age on serum NT-proBNP concentration is significant, with normal levels decreasing with age in children younger than 18 years [15,16]. In the present study, 30.3% (n=99) of all children with sepsis died, and this mortality rate is similar to that reported by a previous meta-analysis [9]. Among the 99 deceased patients, the median serum NT-proBNP levels varied significantly among the 4 age groups (5474 ng/L, 5061 ng/L, 4755 ng/L, and 4435 ng/L for ages of <1 year, 1–3 years, 4–6 years, and 7–18 years, respectively;
Regarding the optimal timing for the collection of venous blood samples for NT-proBNP, there is no consensus. Villalba et al proposed that collection at 72 h after admission is appropriate [6]. Papanikolaou et al recommended that values derived from multiple sample collection times could better predict 28-day mortality than a value obtained at a single time point [22]. However, our results showed that the amount of fluid infused during treatment was much higher in patients with septic shock than in those with another sepsis diagnosis, and a high liquid infusion volume was associated with a significant increase in NT-proBNP release (
In this study, the median level of NT-proBNP in patients with septic shock was significantly higher than that in patients with other forms of sepsis in all of the different age groups (all
In a valuable meta-analysis, serum procalcitonin was shown to have a sensitivity of 77% and specificity of 79% for detecting sepsis [23]. Yu et al showed that serum procalcitonin level could be used to predict sepsis-related mortality [24]. Inthe present study, the serum NT-proBNP level increased with increasing serum procalcitonin, and positive correlations were observed for all age groups (all
The objective of this study was to analyze the ability of the serum NT-proBNP level to predict short-term mortality in children with sepsis and to determine the appropriate age range-specific cut-off values. Our ROC curve analysis for the ability of serum NT-proBNP to predict short-term (28-day) mortality in pediatric sepsis patients of differing age produced age range-specific NT-proBNP cut-off values of 5000 ng/L, 4500 ng/L, 4100 ng/L, and 3800ng/L for sepsis patients aged <1 year, 1–3 years, 4–6 years and 7–18 years, respectively. The corresponding AUC, sensitivity, and specificity values as well as the positive likelihood ratios supported the promising ability of these cut-off values for clinical mortality prediction in these patients. Importantly, these are the reported age range-specific prediction values for children under 18 years of age, and they may be useful for the management of sepsis in pediatric patients.
The present study is limited by being a single-center, retrospective study with a small sample size, and we did not use the quick sequential organ failure assessment (qSOFA) score tool, all of which may have biased the results. Larger dedicated studies in patients from multiple regions as well as analyses of these biomarker ranges in mortality prediction models are needed to confirm whether the NT-proBNP cut-off values we determined are appropriate for use in children worldwide.
Conclusions
The aim of this retrospective study from a single center was to determine age-stratified cut-off values for serum levels of NT-proBNP and mortality from sepsis in children under 18 years. The findings in children stratified by age at <1 year, 1–3 years, 4–6 years, and 7–18 years identified cut-off values that predicted sepsis-associated mortality of 5000 ng/L, 4500 ng/L, 4100 ng/L, and 3800 ng/L, respectively. Larger dedicated studies in patients from multiple regions will help to the determine whether these d NT-proBNP cut-off values are appropriate for use in pediatric patients around the world.
Figures
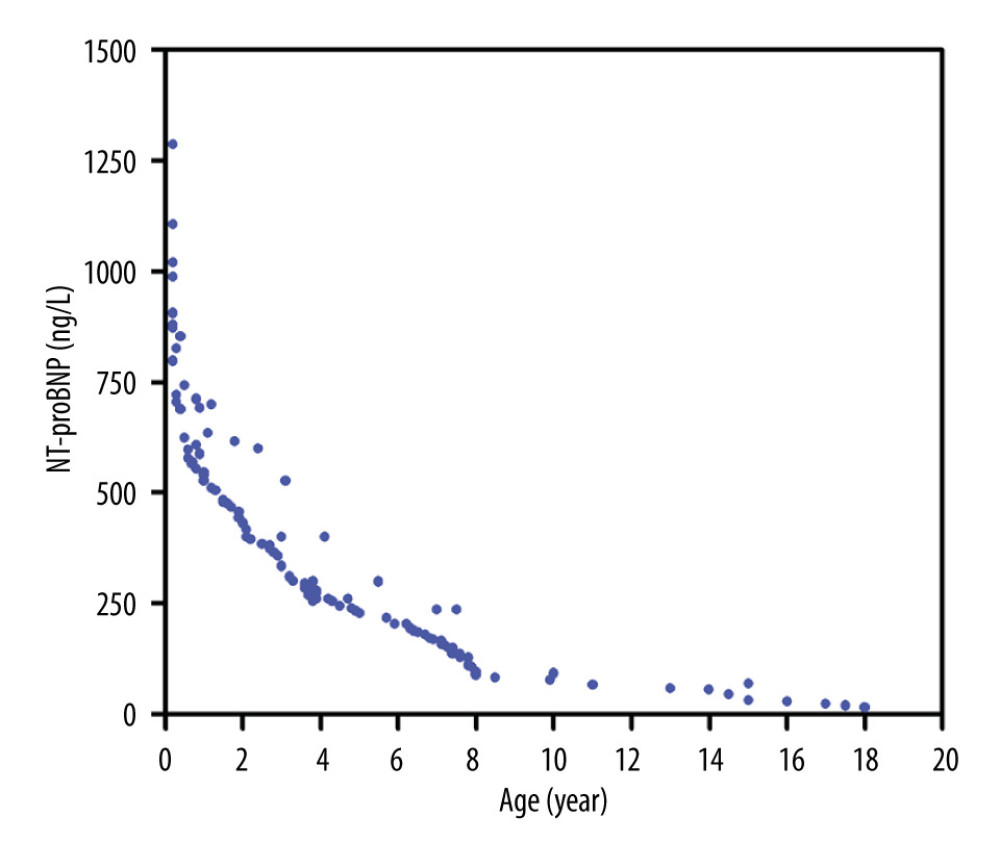 Figure 1. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in healthy children up to age 18. Correlation between the serum NT-proBNP level and age in healthy children was confirmed by values of r=−0.816 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA).
Figure 1. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in healthy children up to age 18. Correlation between the serum NT-proBNP level and age in healthy children was confirmed by values of r=−0.816 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA). 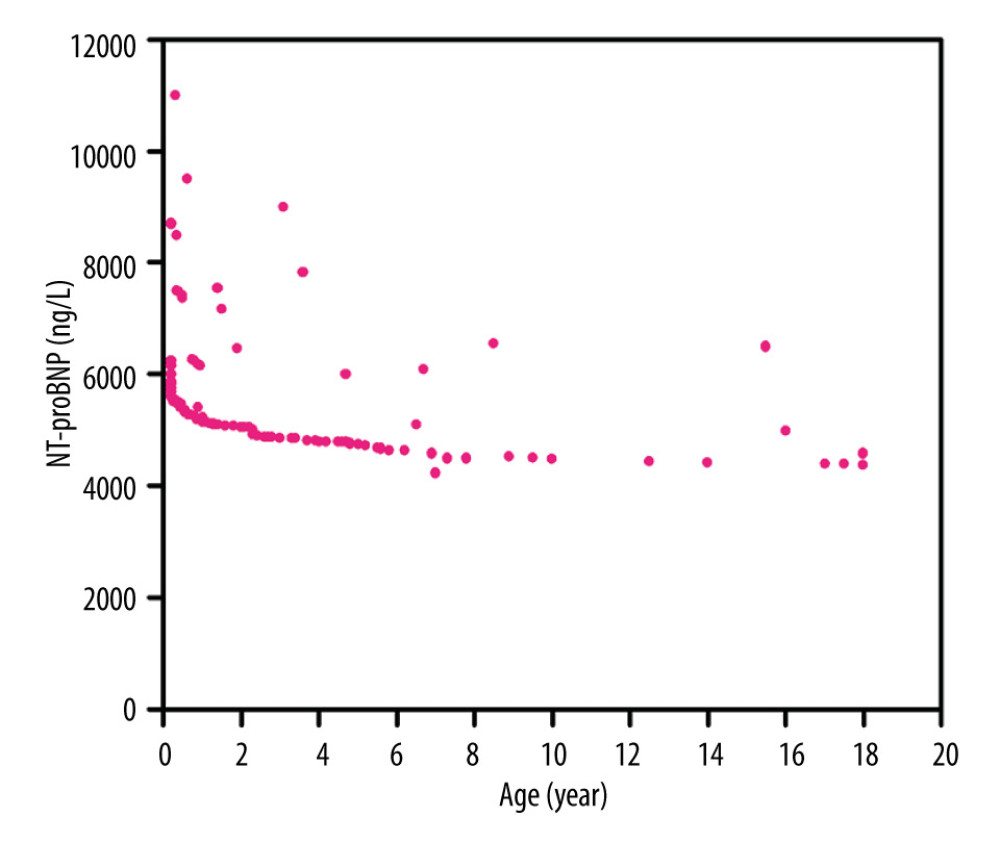 Figure 2. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in pediatric sepsis patients up to age 18. Correlation between the serum NT-proBNP level and age in children with sepsis was confirmed by values of r=−0.325 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA).
Figure 2. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in pediatric sepsis patients up to age 18. Correlation between the serum NT-proBNP level and age in children with sepsis was confirmed by values of r=−0.325 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA). 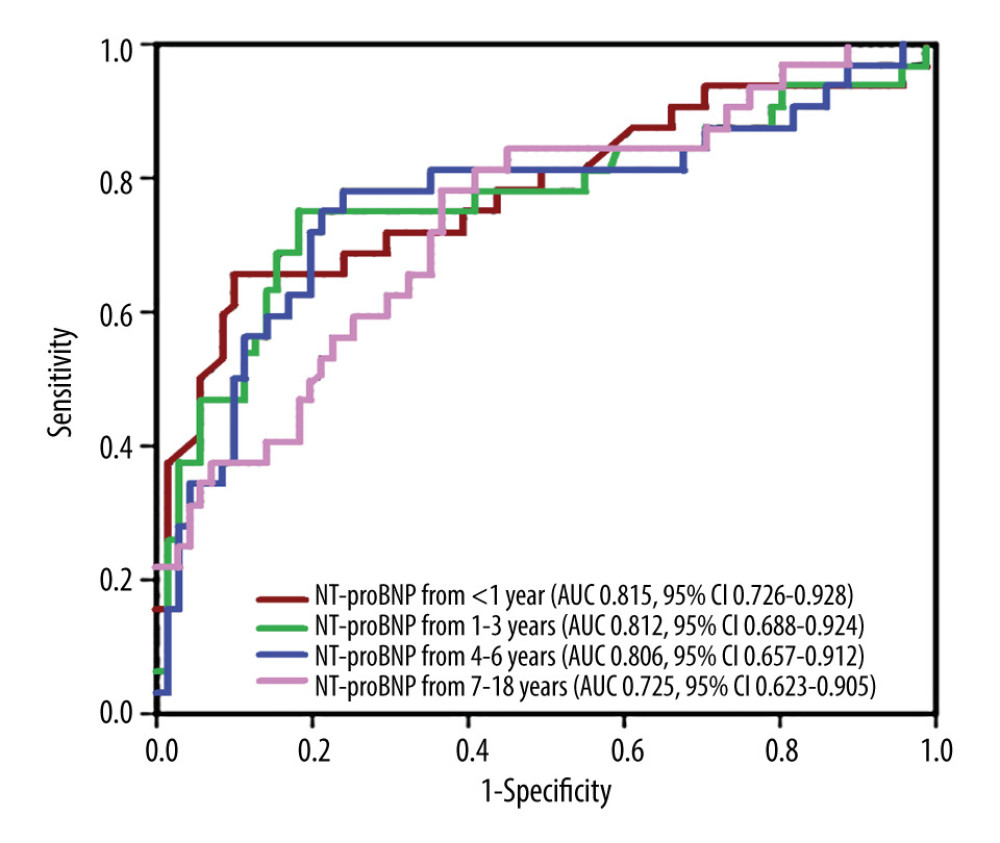 Figure 3. Comparing ROC curves for serum NT-proBNP in patients with sepsis at the ages <1 year, 1–3 years, 4–6 years, And 7–18 years. The AUC with 95% CI values were 0.815(0.73–0.93), 812(0.69–0.92), respectively for ages <1 year, 1–3 years, 4–6 years, and 7–18 years. The sensitivity, specificity, and optimal cut-off values were analyzed, and determining the predictive cut-off value of the serum N-terminal proB-type natriuretic peptide (NT-proBNP) level for 28-day mortality in pediatric sepsis patients of different ages. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA).
Figure 3. Comparing ROC curves for serum NT-proBNP in patients with sepsis at the ages <1 year, 1–3 years, 4–6 years, And 7–18 years. The AUC with 95% CI values were 0.815(0.73–0.93), 812(0.69–0.92), respectively for ages <1 year, 1–3 years, 4–6 years, and 7–18 years. The sensitivity, specificity, and optimal cut-off values were analyzed, and determining the predictive cut-off value of the serum N-terminal proB-type natriuretic peptide (NT-proBNP) level for 28-day mortality in pediatric sepsis patients of different ages. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA). Tables
Table 1. Clinical characteristics of pediatric patients with sepsis at administration.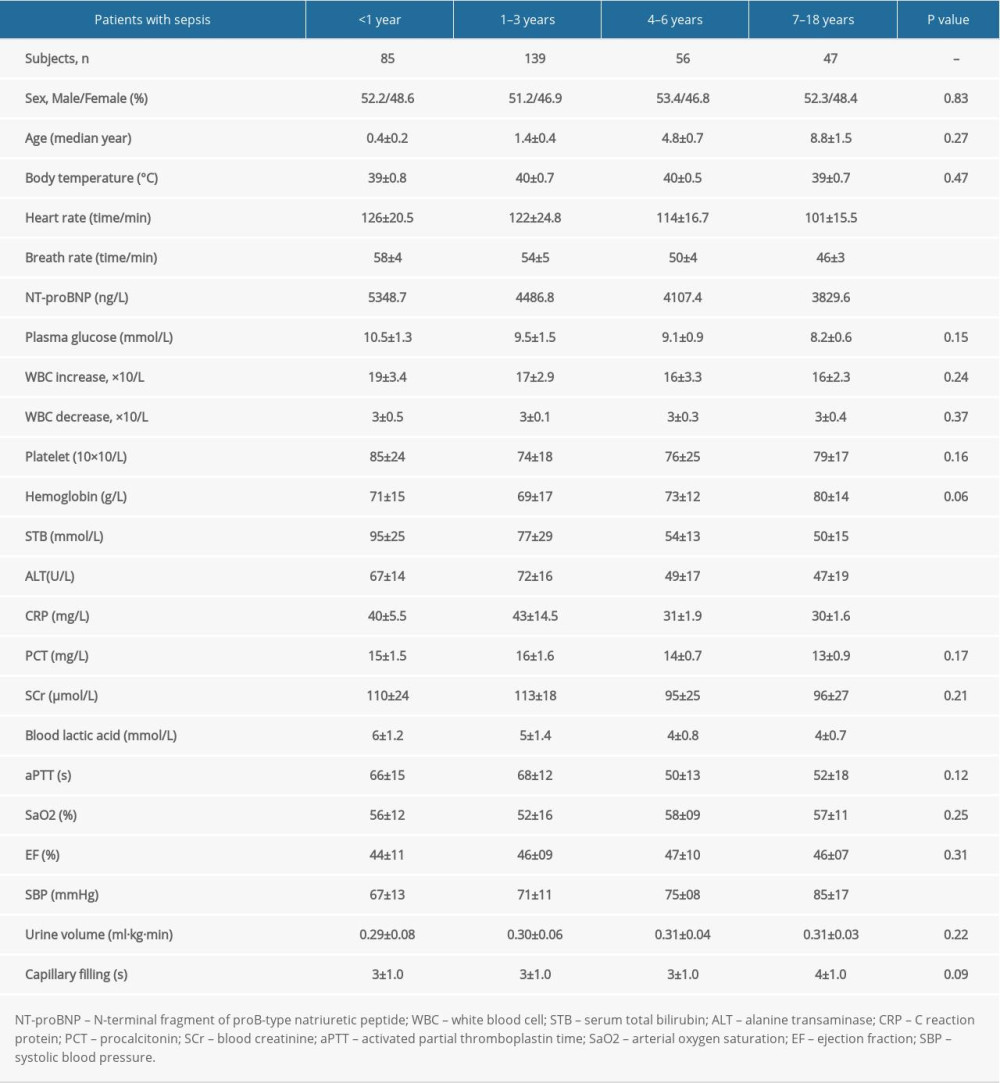 Table 2. Comparison of serum NT-proBNP levels in pediatric sepsis patients of different ages according to the type of septic condition and patient survival.
Table 2. Comparison of serum NT-proBNP levels in pediatric sepsis patients of different ages according to the type of septic condition and patient survival.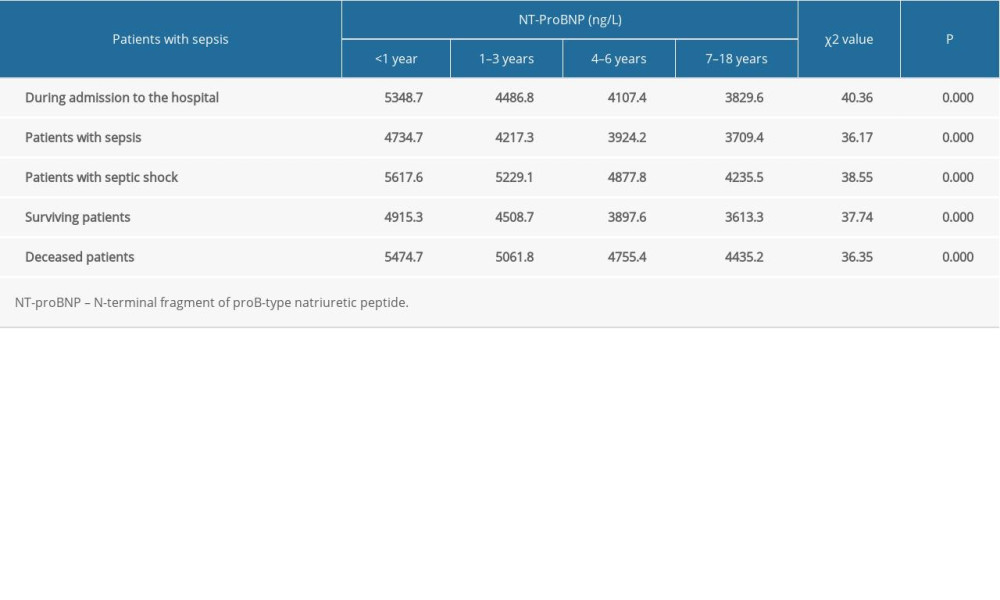 Table 3. Correlation of serum NT-proBNP and procalcitonin levels during admission to the hospital in sepsis patients stratified by age.
Table 3. Correlation of serum NT-proBNP and procalcitonin levels during admission to the hospital in sepsis patients stratified by age.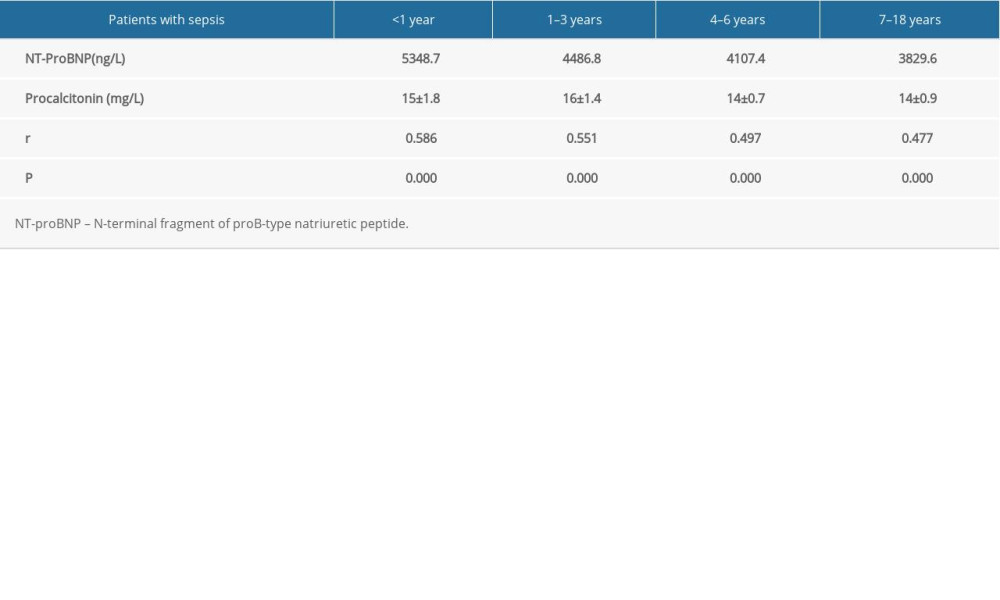 Table 4. ROC analysis of serum NT-proBNP levels for short-term mortality prediction in septic children of different ages.
Table 4. ROC analysis of serum NT-proBNP levels for short-term mortality prediction in septic children of different ages.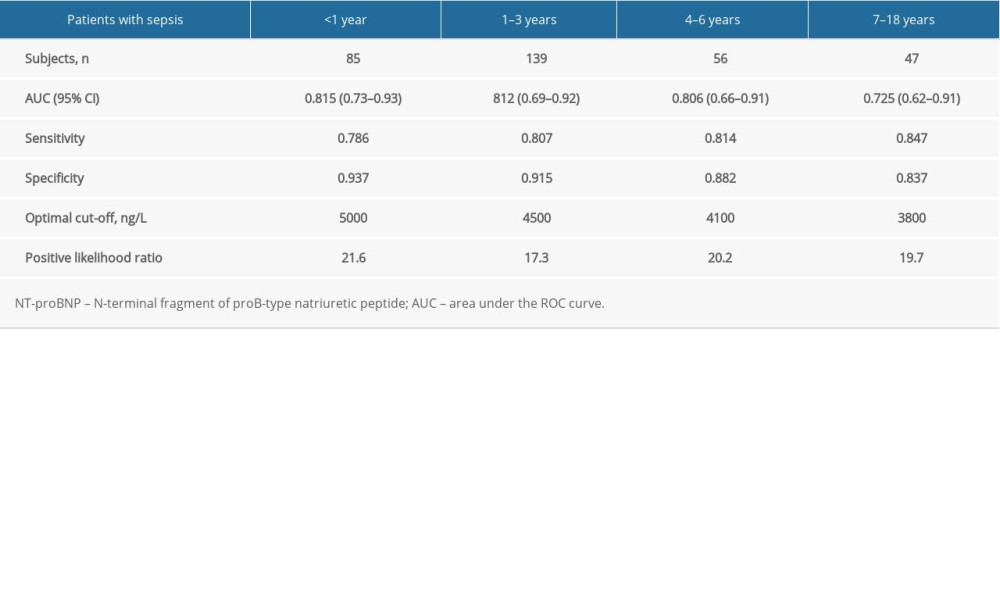
References
1. Saenger AK, Rodriguez-Fraga O, Ler R, Specificity of B-Type natriuretic peptide assays: Cross-reactivity with different BNP, NT-proBNP, and proBNP peptides: Clin Chem, 2017; 63; 351-58
2. Rusconi PG, Ludwig DA, Ratnasamy C, Serial measurements of serum NT-proBNP as markers of left ventricular systolic function and remodeling in children with heart failure: Am Heart J, 2010; 160; 776-83
3. Wong DT, George K, Wilson J, Effectiveness of serial increases in amino-terminal pro-B-type natriuretic peptide levels to indicate the need for mechanical circulatory support in children with acute decompensated heart failure: Am J Cardiol, 2011; 107; 573-78
4. deFilippi CR, de Lemos JA, Tkaczuk AT, Physical activity, change in biomarkers of myocardial stress and injury, and subsequent heart failure risk in older adults: J Am Coll Cardiol, 2012; 60; 2539-47
5. Guaricci AI, Santoro F, Paoletti Perini A, Correlations between NT-proBNP, outcome and haemodynamics in patients with septic shock: Acta Cardiol, 2015; 70; 545-52
6. García Villalba E, Bernal Morell E, Egea MP, The N-terminal pro brain natriuretic peptide is the best predictor of mortality during hospitalization in patients with low risk of sepsis-related organ failure: Med Clin (Barc), 2017; 149; 189-95
7. Chen FC, Xu YC, Zhang ZC, Multi-biomarker strategy for prediction of myocardial dysfunction and mortality in sepsis: J Zhejiang Univ Sci B, 2020; 21; 537-48
8. Alataby H, Nfonoyim J, Diaz K, The levels of lactate, troponin, and N-terminal pro-B-type natriuretic peptide are predictors of mortality in patients with sepsis and septic shock: A retrospective cohort study: Med Sci Monit Basic Res, 2021; 27; e927834
9. Vallabhajosyula S, Wang Z, Murad MH, Natriuretic peptides to predict short-term mortality in patients with sepsis: A systematic review and meta-analysis: Mayo Clin Proc Innov Qual Outcomes, 2020; 4; 50-64
10. Vallabhajosyula S, Deshmukh AJ, Kashani K, Tako-tsubo cardiomyopathy in severe sepsis: Nationwide trends, predictors, and outcomes: J Am Heart Assoc, 2018; 7; e009160
11. Vallabhajosyula S, Sakhuja A, Geske JB, Clinical profile and outcomes of acute cardiorenal syndrome type-5 in sepsis: An eight-year cohort study: PLoS One, 2018; 13; e0190965
12. Vallabhajosyula S, Jentzer JC, Geske JB, New-onset heart failure and mortality in hospital survivors of sepsis-related left ventricular dysfunction: Shock, 2018; 49; 144-49
13. Pandompatam G, Kashani K, Vallabhajosyula S, The role of natriuretic peptides in the management, outcomes and prognosis of sepsis and septic shock: Rev Bras Ter Intensiva, 2019; 31; 368-78
14. Bhandari B, Cunningham J, The role of brain natriuretic peptide as a prognostic marker for sepsis: Cureus, 2020; 12; e8954
15. Deng M, Lin C, Wen T, Zhu H, Yi Z, Plasma N-terminal pro-B-type natriuretic peptide: Selecting the optimal heart failure marker in children of age up to 18 years: International Journal of Clinical & Experimental Medicine, 2016; 9; 10756-62
16. Ross RD, The Ross classification for heart failure in children after 25 years: A review and an age-stratified revision: Pediatr Cardiol, 2012; 33; 1295-300
17. Singer M, Deutschman CS, Seymour CW, The third international consensus definitions for sepsis and septic shock (Sepsis-3): JAMA, 2016; 315; 801-10
18. Chow SL, Maisel AS, Anand I, Role of biomarkers for the prevention, assessment, and management of heart failure: A scientific statement from the American Heart Association: Circulation, 2017; 135; e1054-e91
19. Rauh M, Koch A, Plasma N-terminal pro-B-type natriuretic peptide concentrations in a control population of infants and children: Clin Chem, 2003; 49; 1563-64
20. Gao L, Jiang D, Wen XS, Prognostic value of NT-proBNP in patients with severe COVID-19: Respir Res, 2020; 21; 83
21. Li H, Shan-Shan Z, Jian-Qiang K, Ling Y, Fang L, Predictive value of C-reactive protein and NT-pro-BNP levels in sepsis patients older than 75 years: A prospective, observational study: Aging Clin Exp Res, 2020; 32; 389-97
22. Papanikolaou J, Makris D, Mpaka M, New insights into the mechanisms involved in B-type natriuretic peptide elevation and its prognostic value in septic patients: Crit Care, 2014; 18; R94
23. Wacker C, Prkno A, Brunkhorst FM, Schlattmann P, Procalcitonin as a diagnostic marker for sepsis: a systematic review and meta-analysis: Lancet Infect Dis, 2013; 13; 426-35
24. Yu H, Nie L, Liu A, Combining procalcitonin with the qSOFA and sepsis mortality prediction: Medicine (Baltimore), 2019; 98; e15981
Figures
 Figure 1. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in healthy children up to age 18. Correlation between the serum NT-proBNP level and age in healthy children was confirmed by values of r=−0.816 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA).
Figure 1. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in healthy children up to age 18. Correlation between the serum NT-proBNP level and age in healthy children was confirmed by values of r=−0.816 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA). Figure 2. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in pediatric sepsis patients up to age 18. Correlation between the serum NT-proBNP level and age in children with sepsis was confirmed by values of r=−0.325 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA).
Figure 2. The inverse association between age and serum N-terminal proB-type natriuretic peptide (NT-proBNP) level in pediatric sepsis patients up to age 18. Correlation between the serum NT-proBNP level and age in children with sepsis was confirmed by values of r=−0.325 and P<0.0001. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA). Figure 3. Comparing ROC curves for serum NT-proBNP in patients with sepsis at the ages <1 year, 1–3 years, 4–6 years, And 7–18 years. The AUC with 95% CI values were 0.815(0.73–0.93), 812(0.69–0.92), respectively for ages <1 year, 1–3 years, 4–6 years, and 7–18 years. The sensitivity, specificity, and optimal cut-off values were analyzed, and determining the predictive cut-off value of the serum N-terminal proB-type natriuretic peptide (NT-proBNP) level for 28-day mortality in pediatric sepsis patients of different ages. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA).
Figure 3. Comparing ROC curves for serum NT-proBNP in patients with sepsis at the ages <1 year, 1–3 years, 4–6 years, And 7–18 years. The AUC with 95% CI values were 0.815(0.73–0.93), 812(0.69–0.92), respectively for ages <1 year, 1–3 years, 4–6 years, and 7–18 years. The sensitivity, specificity, and optimal cut-off values were analyzed, and determining the predictive cut-off value of the serum N-terminal proB-type natriuretic peptide (NT-proBNP) level for 28-day mortality in pediatric sepsis patients of different ages. The figure was generated using GraphPad software. (GraphPad Software, La Jolla, USA). Tables
 Table 1. Clinical characteristics of pediatric patients with sepsis at administration.
Table 1. Clinical characteristics of pediatric patients with sepsis at administration. Table 2. Comparison of serum NT-proBNP levels in pediatric sepsis patients of different ages according to the type of septic condition and patient survival.
Table 2. Comparison of serum NT-proBNP levels in pediatric sepsis patients of different ages according to the type of septic condition and patient survival. Table 3. Correlation of serum NT-proBNP and procalcitonin levels during admission to the hospital in sepsis patients stratified by age.
Table 3. Correlation of serum NT-proBNP and procalcitonin levels during admission to the hospital in sepsis patients stratified by age. Table 4. ROC analysis of serum NT-proBNP levels for short-term mortality prediction in septic children of different ages.
Table 4. ROC analysis of serum NT-proBNP levels for short-term mortality prediction in septic children of different ages. Table 1. Clinical characteristics of pediatric patients with sepsis at administration.
Table 1. Clinical characteristics of pediatric patients with sepsis at administration. Table 2. Comparison of serum NT-proBNP levels in pediatric sepsis patients of different ages according to the type of septic condition and patient survival.
Table 2. Comparison of serum NT-proBNP levels in pediatric sepsis patients of different ages according to the type of septic condition and patient survival. Table 3. Correlation of serum NT-proBNP and procalcitonin levels during admission to the hospital in sepsis patients stratified by age.
Table 3. Correlation of serum NT-proBNP and procalcitonin levels during admission to the hospital in sepsis patients stratified by age. Table 4. ROC analysis of serum NT-proBNP levels for short-term mortality prediction in septic children of different ages.
Table 4. ROC analysis of serum NT-proBNP levels for short-term mortality prediction in septic children of different ages. In Press
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








