16 June 2022: Clinical Research
Remimazolam Tosilate Sedation with Adjuvant Sufentanil in Chinese Patients with Liver Cirrhosis Undergoing Gastroscopy: A Randomized Controlled Study
YingHao Cao1ADEF, Ping Chi2AE, Chen Zhou3CD, WenFei Lv2B, ZheFen Quan2BF, Fu Shan Xue1ADEF*DOI: 10.12659/MSM.936580
Med Sci Monit 2022; 28:e936580
Abstract
BACKGROUND: This prospective, randomized, controlled study evaluated the efficacy and safety of remimazolam tosilate sedation with adjuvant sufentanil, relative to propofol, for Chinese patients with liver cirrhosis undergoing gastroscopy.
MATERIAL AND METHODS: Patients with liver cirrhosis (n=148) aged 18-65 years and undergoing gastroscopy were randomly and equally allocated to receive either 0.107 mg/kg remimazolam tosilate (remimazolam group) or 2 mg/kg propofol. Patients received intravenous sufentanil 0.15 μg/kg before the study drug. If necessary, an additional dose of propofol 20 mg was used and repeated. The primary outcome was the satisfaction rating (satisfactory, fair, or unsatisfactory) of the endoscopist with the sedation. Secondary outcomes were complications (respiratory depression, apnea, body movement, bradycardia, hypotension, nausea or vomiting, somnolence, dizziness, and fever) and patient satisfaction.
RESULTS: Compared with the propofol group, the remimazolam group required a longer time to sedation and a shorter time to emergence. The percentage of remimazolam sedations the endoscopist rated satisfactory (90.5%) was higher than that for propofol (77.0%; P=0.026). Patients given remimazolam experienced lower rates of respiratory depression, body movement, and hypotension (2.7, 8.1, 4.1%, respectively), than did the propofol group (17.6, 23.0, 14.9%; P=0.003, 0.013, 0.025). The 2 groups were comparable regarding the other secondary outcomes.
CONCLUSIONS: For Chinese patients with liver cirrhosis undergoing gastroscopy, remimazolam tosilate with adjuvant sufentanil provides a satisfactory level of sedation with a good safety profile.
Keywords: deep sedation, Gastroscopy, Liver Cirrhosis, Remimazolam, Benzodiazepines, China, Humans, Hypnotics and Sedatives, Hypotension, Propofol, Prospective Studies, Respiratory Insufficiency, Sufentanil
Background
Liver cirrhosis is a common cause of mortality, accounting for around 2 million deaths per year globally [1]. Esophagogastric variceal hemorrhage is a life-threatening condition for patients with liver cirrhosis [2]. All patients with cirrhosis should be screened for esophagogastric varices in accordance with the current guidelines, and a standard endoscopy is the preferred for variceal screening and diagnosis [2–4].
Sedation is widely used during gastrointestinal endoscopic procedures. In a recent national survey, the most frequently used sedatives for gastrointestinal endoscopy were propofol, midazolam, and ketamine [5]. In addition, fentanyl, sufentanil, or dezocine are the favored analgesics administered in combination with the sedatives [5]. However, each of these analgesics may cause respiratory depression, and combining propofol or midazolam with opioids further elevates the risk of hypoxemia [6–8]. Patients with liver cirrhosis undergoing gastroscopy require satisfactory sedation with minimal adverse effects.
Remimazolam is a new benzodiazepine that acts upon GABA-A (γ-aminobutyric acid receptor-type A) receptors. Its properties include rapid onset, a short and predictable duration of sedation, and rapid recovery [9]. The maximal sedative effect of remimazolam is attained within 3 minutes of administration, and then it is rapidly metabolized to inactive compounds by tissue esterases [10]. A recent study reported that an ascending dose of remimazolam and then a continuous infusion was safe and well tolerated in healthy volunteers [11]. In addition, the pharmacological effects of remimazolam can be quickly reversed by the specific antagonist flumazenil, which adds to the safety profile of remimazolam. Recent clinical studies suggested that remimazolam provides adequate sedation for gastrointestinal endoscopy or fiberoptic bronchoscopy procedures [12–14]. However, whether remimazolam with adjuvant sufentanil may offer a satisfactory level of sedation for gastroscopy procedures in patients with liver cirrhosis remains unclear [15].
We hypothesized that remimazolam with adjuvant sufentanil would improve the satisfaction of the endoscopist with the sedation, and with fewer associated complications, relative to conventional propofol sedation combined with sufentanil. Thus, the current study investigated the efficacy and safety of remimazolam tosilate sedation, combined with an adjuvant low dose of sufentanil, when administered to patients with liver cirrhosis undergoing gastroscopic procedures. The primary outcome was the rating for satisfaction of the endoscopist with the sedation. The secondary outcomes included sedation-related complications and patient satisfaction.
Material and Methods
STUDY DESIGN:
This investigator-initiated, prospective, randomized, single-blinded, parallel-controlled trial was conducted at Beijing YouAn Hospital Affiliated to Capital Medical University, Beijing, China. The study protocol was approved by the Ethics Committee of Beijing YouAn Hospital on 24 December2020 (approval No. 2020-148). The study was then registered at the Chinese Clinical Trial Registry on 15 January2021 (registration No. ChiCTR2100042179), before the first participant was enrolled. Written informed consent was obtained from each patient.
PATIENTS:
The recruited subjects were aged 18 to 65 years, with American Society of Anesthesiologists (ASA) physical status II to III, Child-Pugh class A, and scheduled for gastroscopy under sedation. The following were criteria for exclusion: history of respiratory or cardiovascular disease; renal dysfunction; known allergy to medications used in this study; inability to understand or complete the survey of satisfaction; or refusal to participate.
RANDOMIZATION AND BLINDING:
An independent biostatistician performed the selection randomization using an online random sequence generator (
SEDATION PROTOCOL:
Patients were fasted overnight, and no sedative medication was given. At approximately 10 to 15 minutes before beginning the gastroscopy procedures, patients received 10 mL of 2% lidocaine jelly (Kangye Pharmaceutical, Handan, Hebei, China) orally for pharyngeal analgesia in a preoperative waiting area. In the endoscopic procedure room, standard monitoring included electrocardiography, non-invasive blood pressure, and peripheral oxygen saturation (SpO2). Patients received nasal oxygen supplementation at a flow of 4 L/min throughout the entire sedation and gastroscopy procedures.
An anesthesia assistant who did not participate in the subsequent study prepared the sedative medications, i.e., remimazolam tosilate (H20190034, Jiangsu Hengrui Medicine, Jiangsu, China) and propofol (H20040079, Guorui Pharmaceutical, Sichuan, China). According to the randomized codes, patients were assigned to receive remimazolam tosilate (remimazolam group) or propofol (propofol group).
The patients in both groups received intravenous sufentanil 0.15 μg/kg with an injection duration of 30 seconds [16].Two minutes later, patients in the remimazolam and propofol groups received, respectively, intravenous remimazolam tosilate 0.107 mg/kg or intravenous propofol 2 mg/kg, with all sedatives delivered within 30 seconds. The dose of remimazolam tosilate was based on our pilot data (unpublished), in which the 95% effective dose of remimazolam (0.107 mg/kg) for gastroscopy in patients with liver cirrhosis was calculated using the Dixon and Massay up-and-down sequential method and probit regression.
When the MOAA/S (Modified Observer Assessment of Alertness and Sedation scale) scoresreached ≤1, the endoscopist performed the gastroscopy using an Olympus CV260 gastroscope system (Olympus Optical, Tokyo, Japan). If the MOAA/S score was greater than 1 or the patient showed clinical signs of an insufficient level of sedation during the procedures (such as body movement, gag reflex, or coughing), rescue sedation with additional propofol at a dose of 20 mg could be used and repeated at the discretion of the attending anesthesiologist.
According to the study protocol, propofol was used as a rescue sedative in both groups. This is because we planned to administer a single medication proven efficacious for rescue sedation in both groups, and propofol is a traditional sedative widely used in gastrointestinal endoscopic procedures.
Patients were transferred to a recovery room after the procedures and stayed for at least 30 minutes. When the patients regained full awareness and were able to walk in a straight line for at least 5 meters without instability, they were permitted to leave the endoscopic unit [17].
Hypotension was defined as a reduction in systolic blood pressure that was 20% or more from the baseline level, and was treated with intravenous ephedrine 5 mg. Bradycardia was defined as a heart rate less than 50 beats per minute, and was treated with intravenous atropine 0.5 mg. Respiratory depression was considered SpO2 less than 90%, and apnea was an absence of breathing lasting 10 seconds or more. Respiratory depression or apnea was treated with airway manipulations (such as chin lift or jaw thrust) or face mask ventilation as necessary.
PRIMARY AND SECONDARY OUTCOMES:
The primary outcome of this study was the level of satisfaction of the endoscopist with the sedation regimen [18], which was rated immediately after each gastroscopy procedure as satisfactory, fair, or unsatisfactory. The secondary outcomes were complications related to the procedure, and patient satisfaction. Complications included respiratory depression, apnea, body movement, bradycardia, hypotension, nausea or vomiting, somnolence, dizziness, and fever. Patient satisfaction was rated as satisfactory, fair, or unsatisfactory.
PERIOPERATIVE DATA:
The collected perioperative data included the following: time to sedation (from drug administration to MOAA/S score ≤1); time to emergence (from the end of the procedures to eye opening); number of patients who needed additional doses of propofol during induction of sedation; number of patients with additional propofol use during the sedation and gastroscopy procedures; and duration of the gastroscopy.
SAMPLE SIZE ESTIMATION:
The number of patients needed in this study was calculated based on our pilot study (not yet published). Our institutional pilot data suggested that endoscopists rated sedation for gastroscopy procedures via remimazolam and propofol as satisfactory in91.7% and 72.5%of procedures, respectively. To detect such a difference between the 2 groups, 118 patients (59 patients in each group) were required, with a power of 80% at an alpha level of 0.05. To compensate a possible dropout rate of 20%, we finally recruited 148 patients, with 74 patients in each group. The sample size estimation was performed using SPSS software (version 19.0; IBM SPSS, Chicago, IL, USA).
STATISTICAL ANALYSIS:
The normal distribution of continuous data was tested using the Kolmogorov-Smirnov test. The continuous data are presented as mean±standard deviation, and compared using the independent t-test. The categorical data are shown as number of patients and percentage, n (%), and were compared using the chi-squared test or Fisher’s exact test. All statistical analyses were performed using SPSS software (version 11.0; IBM SPSS, Chicago, IL, USA). A 2-sided
Results
PATIENT CHARACTERISTICS:
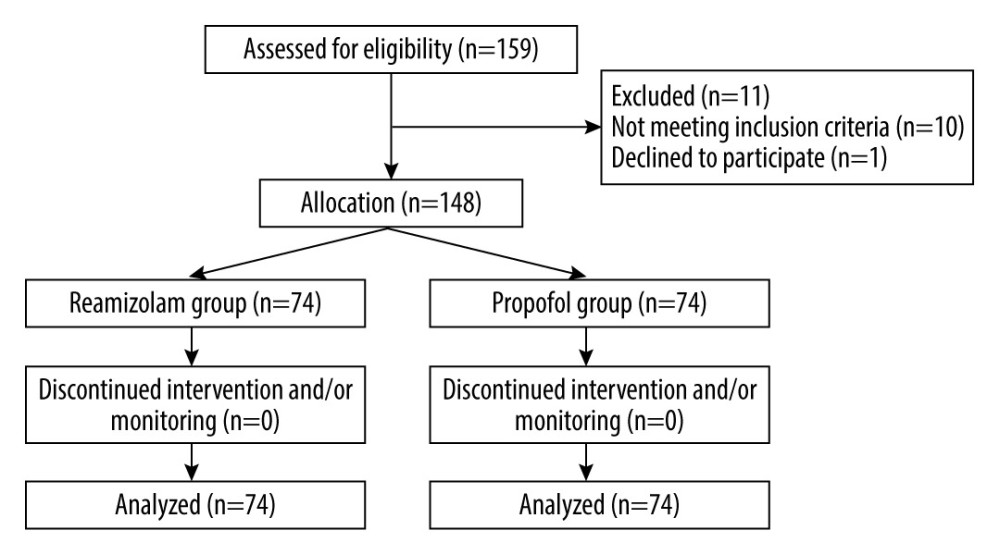
Between 21 February2021 and1 September2021, 159 patients with liver cirrhosis scheduled for gastroscopy under sedation were screened for eligibility for this study (Figure 1). Of these, 11 patients were excluded due to not meeting the inclusion criteria or refusal to participate. Finally, 148 patients were included and randomly and equally assigned to the treatment groups (i.e., 74 patients in each group). Each of the 148 patients completed their gastroscopic procedures with sedation successfully, without dropouts.
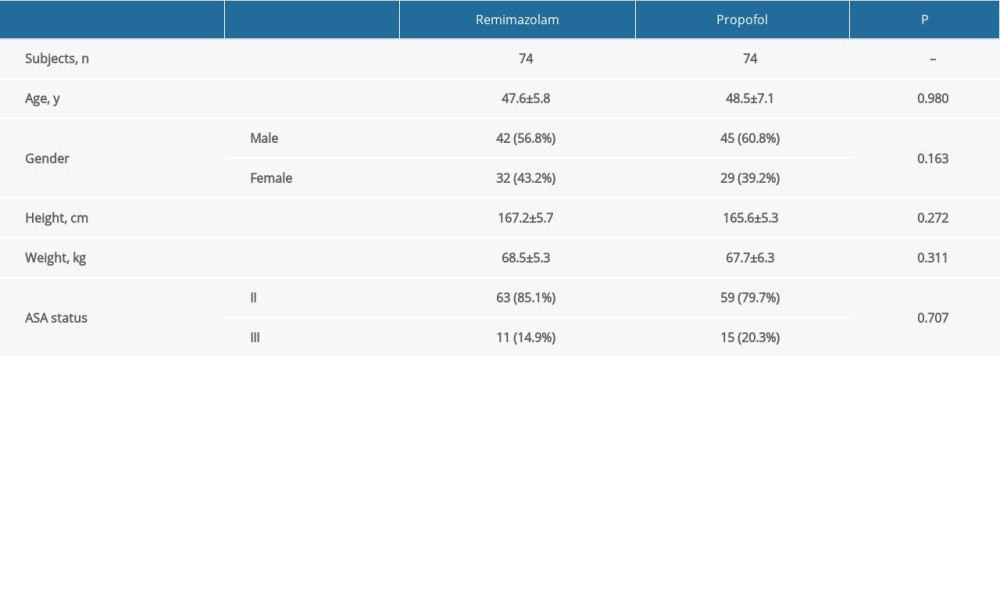
The 2 groups are comparable in terms of patient characteristics (Table 1). The ages of the remimazolam and propofol groups were 47.6±5.8 years and 48.5±7.1 years, respectively. There were 42 (56.8%) and 45 (60.8%) men in the remimazolam and propofol groups. Most patients were at ASA status II (specifically, 85.1% and 79.7% given remimazolam and propofol).
PERIOPERATIVE DATA:
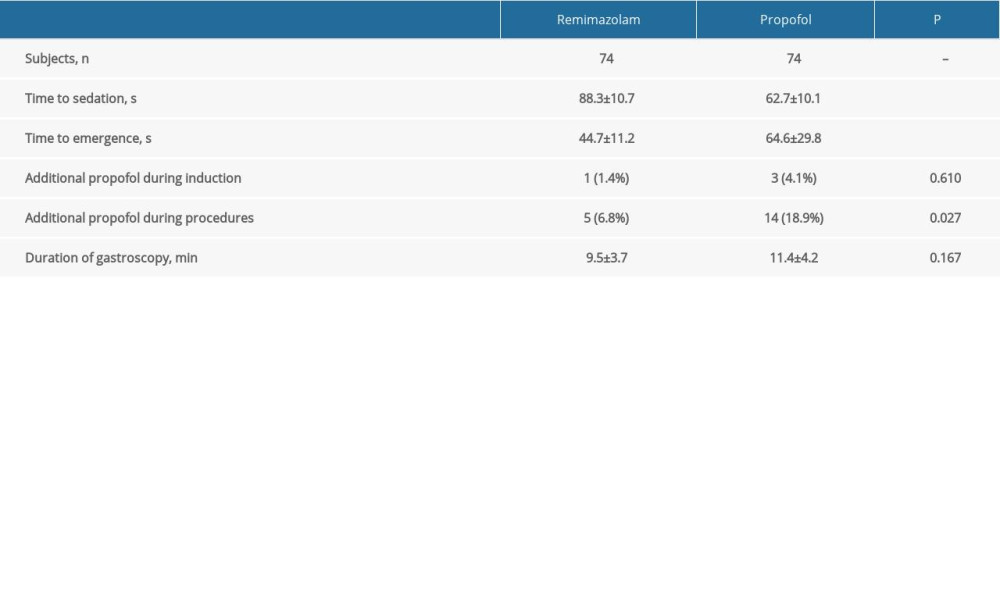
The time to sedation of the remimazolam group (88.3±10.7 s) was longer than that of the propofol group (62.7±10.1 s; P<0.001), while the time to emergence from sedation of the remimazolam group (44.7±11.2 s) was less than that of the propofol group (64.6±29.8 s; P<0.001; Table 2). One patient (1.4%) and 3 patients (4.1%) in the remimazolam and propofol groups, respectively, needed additional doses of propofol during induction of sedation. Five (6.8%) and 14 patients (18.9%) in the remimazolam and propofol groups received additional propofol doses during the sedation and gastroscopy procedures (P=0.027). The duration of gastroscopy was 9.5±3.7 minutes and 11.4±4.2 minutes in the remimazolam and propofol groups.
PRIMARY OUTCOME:
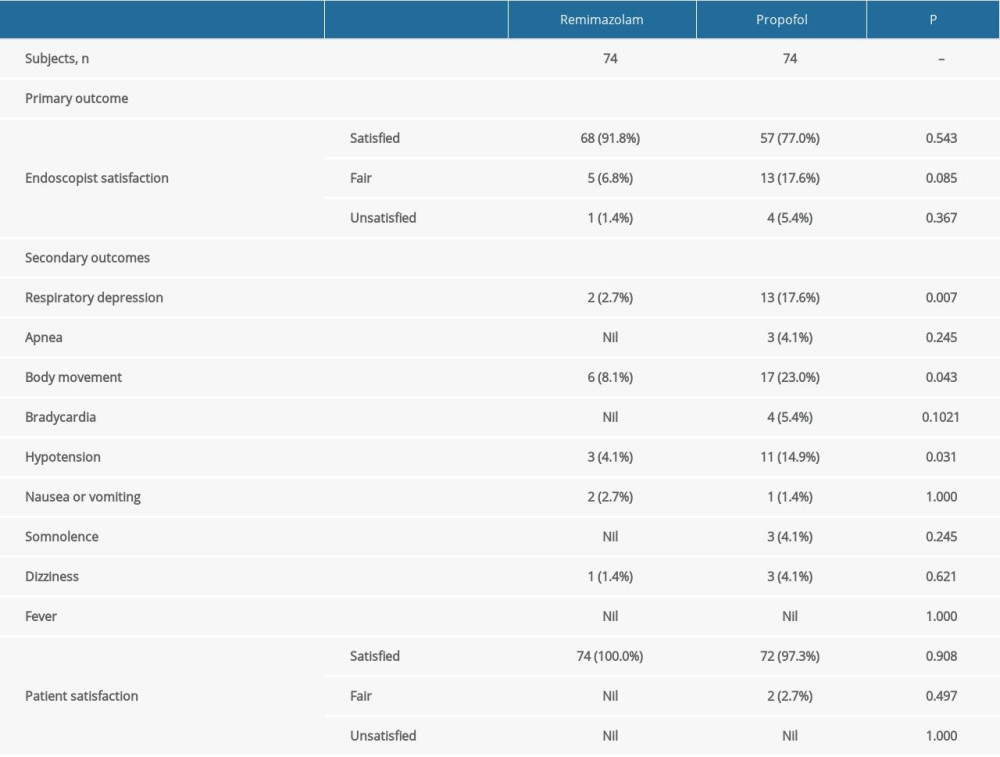
The endoscopist rated the sedation process as satisfactory in 67 of 74 (90.5%) of the procedures in the remimazolam group, which was significantly higher than that of the 57 of 74 (77.0%) procedures in the propofol group (P=0.026; Table 3).
SECONDARY OUTCOMES:
A significantly lower number of patients in the remimazolam group developed respiratory depression (2.7%), body movement (8.1%), or hypotension (4.1%), when compared with the propofol group (17.6%, 23.0%, 14.9%; P=0.003, 0.013, 0.025; Table 3). There were no between-group differences in the other secondary outcomes, including apnea, bradycardia, nausea or vomiting, somnolence, dizziness, fever, or patient satisfaction.
Discussion
MAIN FINDINGS OF THIS TRIAL:
To the best of our knowledge, this is the first randomized controlled trial to show the efficacy and safety of remimazolam tosilate sedation, with an adjuvant low dose of sufentanil, in patients with liver cirrhosis undergoing gastroscopic procedures. The results indicate that remimazolam tosilate combined with adjuvant sufentanil provides adequate and satisfactory sedation for gastroscopy in these patients. Compared with propofol, the use of remimazolam led to higher endoscopist satisfaction with the sedation regimen, and fewer incidences of perioperative respiratory depression, body movement, or hypotension. Fewer patients in the remimazolam group than in the propofol group needed additional doses of propofol during the procedures. In addition, patients receiving remimazolam combined with adjuvant sufentanil experienced a longer time to sedation and a shorter time to emergence, than did patients receiving propofol and sufentanil.
COMPARISONS WITH THE EXISTING LITERATURE:
As a novel benzodiazepine drug characterized by ultra-short sedative and hypnotic effects, remimazolam has been introduced into clinical practice for sedation and general anesthesia. A phase IIb clinical study reported that the use of remimazolam produced an adequate level of sedation for patients undergoing colonoscopy, as reflected by the high rate of successful sedation and a good safety profile [12]. Later, a phase III clinical trial found that patients receiving remimazolam for colonoscopy administered by endoscopists experienced a safer sedation and a faster neuropsychiatric recovery than did those given midazolam [19]. Another study reported that remimazolam provided safe and effective sedation during flexible bronchoscopy [20]. The results of a recent multicenter, randomized, phase III trial showed that the rate of sedation success of remimazolam tosilate was not inferior to propofol in gastrointestinal endoscopy, with a favorable safety profile and a quick recovery from sedation [13]. Remimazolam was well tolerated by surgical patients and was also not inferior to propofol for general anesthesia induction and maintenance [21]. Given that patients with liver cirrhosis have an increased susceptibility to sedative drugs, a tailored pharmacotherapy is needed to ensure safety [22,23]. In this regard, remimazolam may be a suitable alternative to conventional sedative agents for these patients.
CLINICAL IMPLICATIONS OF OUR RESULTS:
The results of the present study may be used to advocate the clinical application of our sedation regimen for patients with liver cirrhosis undergoing gastroscopic procedures. The combination of remimazolam tosilate with a low dose of adjuvant sufentanil for sedation and analgesia in these patients led to a high rate of endoscopist satisfaction and successful completion of the procedures. Furthermore, a significantly lower number of patients in the remimazolam group developed respiratory depression, body movement, or hypotension compared with the propofol group. No severe adverse events associated with remimazolam sedation were observed. This lack of complications is extremely important for patients with liver cirrhosis who are already at an increased risk for postoperative morbidity.
LIMITATIONS OF THIS STUDY:
There are several limitations in this study. First, the attending anesthesiologist was not blinded to the group allocation, because the obvious difference in the appearance of remimazolam and propofol made blinding unfeasible. The non-blinding may have confounded the current results. Second, only patients with liver cirrhosis aged 18 to 65 years, with ASA classification II–III and Child-Pugh class A were included in this study. This is because we believe that sedation for older patients or with higher ASA or Child-Pugh classes scheduled for gastroscopy can reference the current results. Last, patients were not followed-up after hospital discharge. Overall, results of this study warrant verification in larger clinical studies of patients with liver cirrhosis undergoing gastroscopic procedures.
Conclusions
This randomized controlled trial suggests that remimazolam tosilate combined with adjuvant sufentanil is a better alternative to propofol with sufentanil for sedation in gastroscopy in Chinese patients with liver cirrhosis. The endoscopist rated the former more highly for satisfaction with sedation, and there were fewer incidences of respiratory depression, body movement, and hypotension.
References
1. Asrani SK, Devarbhavi H, Eaton J, Burden of liver diseases in the world: J Hepatol, 2019; 70; 151-71
2. Tsochatzis EA, Bosch J, Burroughs AK, Liver cirrhosis: Lancet, 2014; 383; 1749-61
3. Xu XY, Ding HG, Li WG, Chinese guidelines on the management of liver cirrhosis (abbreviated version): World J Gastroenterol, 2020; 26; 7088-103
4. Tripathi D, Stanley AJ, Hayes PC, guidelines on the management of variceal haemorrhage in cirrhotic patients: Gut, 2015; 64; 1680-704
5. Zhou S, Zhu Z, Dai W, National survey on sedation for gastrointestinal endoscopy in 2758 Chinese hospitals: Br J Anaesthesia, 2021; 127; 56-64
6. American Society of Anesthesiologists Task Force on Sedation and Analgesia by Non-Anesthesiologists, Practice guidelines for sedation and analgesia by non-anesthesiologists: Anesthesiology, 2002; 96; 1004-17
7. Zhang W, Zhu Z, Zheng Y, Effect and safety of propofol for sedation during colonoscopy: A meta-analysis: J Clin Anesth, 2018; 51; 10-18
8. Friedrich-Rust M, Welte M, Welte C, Capnographic monitoring of propofol-based sedation during colonoscopy: Endoscopy, 2014; 46; 236-44
9. Antonik LJ, Goldwater DR, Kilpatrick GJ, A placebo- and midazolam-controlled phase I single ascending-dose study evaluating the safety, pharmacokinetics, and pharmacodynamics of remimazolam (CNS 7056): Part I. Safety, efficacy, and basic pharmacokinetics: Anesth Analg, 2012; 115; 274-83
10. Wiltshire HR, Kilpatrick GJ, Tilbrook GS, A placebo- and midazolam-controlled phase I single ascending-dose study evaluating the safety, pharmacokinetics, and pharmacodynamics of remimazolam (CNS 7056): Part II. Population pharmacokinetic and pharmacodynamic modeling and simulation: Anesth Analg, 2012; 115; 284-96
11. Sheng XY, Liang Y, Yang XY, Safety, pharmacokinetic and pharmacodynamic properties of single ascending dose and continuous infusion of remimazolam besylate in healthy Chinese volunteers: Eur J Clin Pharmacol, 2020; 76; 383-91
12. Pambianco DJ, Borkett KM, Riff DS, A phase IIb study comparing the safety and efficacy of remimazolam and midazolam in patients undergoing colonoscopy: Gastrointest Endosc, 2016; 83; 984-92
13. Chen SH, Yuan TM, Zhang J, Remimazolam tosilate in upper gastrointestinal endoscopy: A multicenter, randomized, non-inferiority, phase III trial: J Gastroenterol Hepatol, 2021; 36; 474-81
14. Jia Z, Ren LX, Fan YTObservation of effective dosage of remimazolam tosilate used for moderate-to-deep sedation in fiberoptic bronchoscopy: Zhonghua Yi Xue Za Zhi, 2021; 101; 813-16 [in Chinese]
15. Dossa F, Megetto O, Yakubu M, Sedation practices for routine gastrointestinal endoscopy: A systematic review of recommendations: BMC Gastroenterol, 2021; 21(1); 22
16. Zhang L, Bao Y, Shi D, Comparing the pain of propofol via different combinations of fentanyl, sufentanil or remifentanil in gastrointestinal endoscopy: Acta Cir Bras, 2014; 29(10); 675-80
17. Sato M, Horiuchi A, Tamaki M, Safety and effectiveness of nurse-administered propofol sedation in outpatients undergoing gastrointestinal endoscopy: Clin Gastroenterol Hepatol, 2019; 17; 1098-104e1091
18. Lin OS, Kozarek RA, Tombs D, The first US clinical experience with computer-assisted propofol sedation: A retrospective observational comparative study on efficacy, safety, efficiency, and endoscopist and patient satisfaction: Anesth Analg, 2017; 125(3); 804-11
19. Rex DK, Bhandari R, Desta T, A phase III study evaluating the efficacy and safety of remimazolam (CNS 7056) compared with placebo and midazolam in patients undergoing colonoscopy: Gastrointest Endosc, 2018; 88; 427-37e426
20. Pastis NJ, Yarmus LB, Schippers F, Safety and efficacy of remimazolam compared with placebo and midazolam for moderate sedation during bronchoscopy: Chest, 2019; 155; 137-46
21. Doi M, Morita K, Takeda J, Efficacy and safety of remimazolam versus propofol for general anesthesia: A multicenter, single-blind, randomized, parallel-group, phase IIb/III trial: J Anesth, 2020; 34; 543-53
22. Weersink RA, Burger DM, Hayward KL, Safe use of medication in patients with cirrhosis: Pharmacokinetic and pharmacodynamic considerations: Expert Opin Drug Metab Toxicol, 2020; 16; 45-57
23. Tsai HC, Lin YC, Ko CL, Propofol versus midazolam for upper gastrointestinal endoscopy in cirrhotic patients: A meta-analysis of randomized controlled trials: PLoS One, 2015; 10; e0117585
Errate
In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952