21 June 2022: Clinical Research
Prognostic Significance of Preoperative Integrated Liver Inflammatory Score in Patients with Hepatocellular Carcinoma
Biao Wu12ABCDEF, Yumeng WuDOI: 10.12659/MSM.937005
Med Sci Monit 2022; 28:e937005
Abstract
BACKGROUND: The Integrated Liver Inflammatory Score (ILIS), which includes 5 serum indicators (albumin, bilirubin, neutrophil count, alpha-fetoprotein [AFP], and alkaline phosphatase [ALP]), is a novel inflammation-based predictive model associated with poor survival in hepatocellular carcinoma (HCC) patients. Our study aimed to assess the prognostic value of ILIS in HCC patients undergoing radical hepatectomy and establish a nomogram and artificial neural network based on their ILIS scores.
MATERIAL AND METHODS: This multicenter retrospective study included patients from 2 institutions from 2007 to 2017. Independent risk factors associated with Recurrence-free survival (RFS) and overall survival (OS) were identified through univariate and multifactor analysis in the training and validation groups, respectively. Afterward, column line graphs and artificial neural networks (ANN) were constructed and validated using the validation group.
RESULTS: A total of 432 patients were included in this study (275 in the training group and 157 in the validation group). In both cohorts, ILIS was correlated with pathological features such as tumor size, degree of differentiation, Child-Pugh class classification, and BCLC staging. Moreover, ILIS was identified as an independent risk factor for OS. ILIS-based nomograms and artificial neural networks also showed the prognostic value of ILIS.
CONCLUSIONS: Preoperative ILIS is an independent and effective predictor of prognosis in HCC patients treated with radical hepatectomy, as shown by the fact that higher ILIS are associated with worse patient prognosis. We have also established nomograms and ANNs that predict HCC prognosis with high accuracy.
Keywords: Prognosis, hepatocellular carcinoma, Integrated Liver Inflammatory Score, Carcinoma, Hepatocellular, Hepatectomy, Hepatitis, Humans, Liver Neoplasms
Background
Liver cancer is the sixth most common cancer worldwide and the third leading cause of cancer deaths worldwide in 2020 [1], with hepatocellular carcinoma (HCC) accounting for 90% of primary liver cancer cases. Unlike in Western countries and regions, most HCC patients in China have a background of hepatitis B virus infection, resulting in cirrhosis [2], with surgery being the first-line treatment for patients with early to mid-stage HCC [3]. Although treatment techniques and perioperative care continue to improve, the overall prognosis is still poor due to the high recurrence and metastasis rates after hepatectomy [4]. Therefore, the construction and validation of highly accurate and sensitive preoperative biomarkers that identify high-risk patients with liver cancer recurrence and assess their prognosis are necessary to improve the clinical prognosis of patients with liver cancer.
An increasing number of studies have shown that the albumin-bilirubin (ALBI) score [5] and the albumin-platelet (PAL) score [6] to be prognostic models for HCC. In addition, preoperative albumin-indocyanine green evaluation (ALICE) scores for HCC patients can predict risk of postoperative liver failure and death [7]. Recently, Chan et al proposed a new predictive model, the ILIS, which can be used to assess the prognosis of patients with liver cancer [8]. This model not only includes inflammatory indicators such as albumin and neutrophils, but also liver cancer-specific indicators such as AFP and ALP, both of which are not included in other models. This feature allows ILIS to perform better than other inflammatory indicators in prognosis assessment. Recent studies have shown that ILIS can predict outcomes in HCC patients after transarterial chemoembolization (TACE) [9]. However, the efficacy of ILIS as a predictive marker is not yet established in patients with early to mid-stage HCC who are amenable to radical resection. Therefore, our study aimed to explore the prognostic significance of ILIS in preoperative patients with early to mid-stage HCC and to develop a corresponding nomogram and ANN model.
Material and Methods
PATIENT CRITERIA:
This study included patients who underwent initial treatment with radical hepatectomy at 2 hospitals from February 2007 to August 2017. This study was approved by the institutional review boards of the 2 participating hospitals, and all participants provided written informed consent. All procedures were conducted in accordance with the Declaration of Helsinki. HCC diagnosis was based on the diagnostic criteria of the American Association for the Study of Liver Diseases and the European Association for the Study of the Liver [10,11]. Patients from the Affiliated Tumor Hospital of Nantong University were included in the training group and patients from the Nantong University Hospital were included in the validation group.
Patients underwent routine investigations, including hematological and biochemical profile, AFP levels, chest X-ray, abdominal ultrasound and computed tomography (CT), or magnetic resonance imaging (MRI) within 7 days before surgery. Patients who met the following criteria were included in this study: (1) no other tumors were present, (2) no preoperative anti-tumor treatment (eg, TACE, radiofrequency ablation, chemotherapy, liver transplantation), (3) postoperative pathological diagnosis of liver cancer, (4) no vital perioperative organ dysfunction, and (5) complete clinical examination reports and follow-up information.
TREATMENT AND FOLLOW-UP:
The patients were treated with radical hepatectomy, meaning that the tumor was considered completely removed macroscopically and histologically. After surgery, all patients were followed up every 2 months for the first year and every 6 months after that. Routine hematology and imaging were performed at each follow-up visit. Once HCC recurrence was suspected, CT or MRI was performed immediately to confirm the diagnosis. Patients with recurrence were treated with modalities such as surgery, TACE, ablation, and chemotherapy, depending on the general condition and tumor status. RFS and OS were calculated from the date of surgery to the date of detection of recurrence and death or last follow-up.
ILIS ASSESSMENT:
ILIS involves 5 serum markers (albumin, bilirubin, neutrophil count, AFP, and ALP), and is calculated as:
Serum data were obtained via examination within 7 days before surgery, with the patients not receiving any anti-inflammatory treatment prior to surgery. The ROC (receiver operating characteristic) curve was used to determine the cut-off value for ILIS based on the patient’s survival status. Patients with HCC were staged using the BCLC (Barcelona Clinical Liver Cancer) system [12].
NOMOGRAMS AND ANN MODELING:
The construction of the nomogram and ANN relied on significant risk factors obtained from univariate and multifactorial analyses. We used the consistency C statistic (C-index) to assess the predictive power of the nomogram. The calibration plots were able to verify the predicted performance of our nomograms at 3 and 5 years after surgery. The ANN was constructed by randomly selecting two-thirds of the patients in the training cohort and cross-validating it with the rest of the cohort. The cross-validation process guarantees the stability and practicability of artificial neural networks [13]. In this study, a 3-layer feed-forward neural network was constructed with input nodes, hidden layers, and output nodes. The ANN has an input layer, an output layer, and multiple hidden layers. By varying the connection weights between neurons, the ANN model learns to associate each input with the corresponding output.
STATISTICAL ANALYSIS:
Statistical analyses were performed using GraphPad Prism software (La Jolla, CA, USA) and SPSS 26.0 (SPSS Company, Chicago, IL, USA). Quantitative values were analyzed using the
Results
ILIS ASSESSMENT:
A total of 432 patients (training group=275; validation group=157) met the inclusion criteria and were included in the study (Figure 1). According to the ROC curves, the ideal cut-off values for HCC patients in the 2 hospitals were 2.13 and 2.52, respectively. The area under the curve (AUC) of the ILIS was 0.59 (95% CI for the area between 0.52 to 0.66) and 0.60 (95% CI for the area between 0.51 to 0.69), respectively. The cut-off values for ILIS corresponded to sensitivity values of 70% and 51% and specificity values of 48% and 72%, respectively (Figure 2).
RELATIONSHIP BETWEEN PREOPERATIVE ILIS AND CLINICOPATHOLOGICAL FEATURES IN HCC PATIENTS:
As shown in Table 1, we analyzed the relationship between the preoperative ILIS and clinicopathological characteristics of HCC patients in both cohorts. In the training group, 166 (60.36%) patients comprised the high ILIS group (ILIS >2.13) while the remaining 109 (39.64%) patients comprised the low ILIS group (ILIS <2.13). Preoperative ILIS was strongly correlated with tumor size, tumor differentiation, Child-Pugh class, and BCLC stage (P<0.05), but not with age, sex, hepatitis, cirrhosis, number of tumors, and vascular cancer plugs (p>0.05). In the validation group, 61 (38.85%) patients were included in the high ILIS group (ILIS >2.52), and 96 (61.15%) were in the low ILIS group (ILIS <2.52). Unlike in the training group, there was no significant correlation between preoperative ILIS and tumor size in this group of patients (P>0.05).
CORRELATION BETWEEN PREOPERATIVE ILIS AND POSTOPERATIVE OS AND RFS IN HCC PATIENTS:
The Kaplan-Meier survival curves for both the training and validation groups showed that the high expression of preoperative ILIS was associated with shorter OS and RFS in HCC patients (Figure 3).
Using univariate analysis, we found that preoperative ILIS, tumor size, tumor number, vascular tumor thrombus, and BCLC stage were all associated with postoperative OS in HCC patients in both cohorts (P<0.05), with the degree of tumor differentiation also associated with OS in the training group (P<0.05) (Table 2). Other clinical characteristics such as age, gender, and cirrhosis were not significantly correlated with OS (P>0.05). Multivariate analysis showed that preoperative ILIS and BCLC stage were independent risk factors for OS in patients with HCC (P<0.05).
In addition, univariate analysis showed that preoperative ILIS, tumor size, vascular tumor thrombus, BCLC stage, and cirrhosis were associated with RFS in patients (P<0.05) (Table 3). However, multivariate analysis showed that only the BCLC stage was an independent risk factor for RFS (P<0.05), unlike ILIS (P>0.05).
NOMOGRAM AND ANN MODELING:
Several important clinicopathological features of each patient were scored according to a columnar chart and the sum of the points obtained was used to accurately predict patient survival (Figure 4A). The higher the score, the worse the prognosis. The model showed high accuracy in predicting the OS of HCC patients after hepatectomy, with a c-index of 0.563. The calibration plots also showed good predictions for the 3- and 5-year OS (Figure 4B, 4C).
Similarly, the ANN model related to the preoperative ILIS score was developed based on several important risk factors (Figure 5A). The proportion of OS importance accounted for by risk factors such as BCLC stage, ILIS, vascular cancer thrombus, and tumor size in the artificial neural network model were 0.272, 0.242, 0.185, and 0.164, respectively (Figure 5B).
Discussion
Surgical resection remains the principal treatment for early to mid-stage HCC. Radical hepatectomy is preferred when preoperative hematology and imaging show that the patient’s liver is in good condition and that the tumor is resectable under current technical conditions [14]. However, the prognosis of patients with HCC varies from patient to patient and depends on tumor status and liver function [15]. Therefore, it is crucial to predict the patient’s prognosis and guide postoperative treatment to increase their chances of survival.
Many studies have shown that inflammation can contribute to the development and progression of cancer through a variety of mechanisms, such as cancer cell proliferation, angiogenesis [16], and genetic mutations [17]. In China, most patients with HCC have hepatitis, and those who do not receive antiviral treatment are more likely to develop liver cancer [18]. As a result, hematological indicators reflecting systemic inflammation are receiving increasing attention, and these indicators are being combined to build prognostic models that can assess patient survival [18]. For instance, the PAL score can be used to predict perioperative survival in HCC patients who undergo hepatectomy [19], and the ALBI score, which is used to assess the prognosis of HCC patients treated with TACE [20]. In addition, the HCC-specific indicator AFP is also considered highly valuable in the diagnosis and prognosis assessment of HCC patients [21,22]. The establishment of a new inflammation-based indicator for HCC, ILIS, is a promising, novel approach for predicting HCC prognosis. Chan et al conducted a retrospective study in over 1000 patients and found that the ILIS was more accurate than other conventional inflammatory indicators such as NLR. It was observed in that study that PLR alone was more accurate in assessing the prognosis of patients, compared with. With an emphasis on health and the accessibility of medical screening, more patients with HCC are being diagnosed in the early to mid-stage. Therefore, we aimed to demonstrate the validity of preoperative ILIS as a potential prognostic indicator for HCC patients. Specifically, we wanted to develop an ILIS-based model that predicts the prognosis of patients with early to mid-stage HCC undergoing radical hepatectomy.
This study retrospectively analyzed HCC patients undergoing surgery and divided them into training and control groups. The optimal threshold values of preoperative ILIS were first determined based on the ROC curves, with the greatest joint sensitivity and specificity. Next, we analyzed the correlation between the preoperative ILIS and clinicopathological characteristics of the patients. The results showed that an elevated ILIS was positively correlated with tumor size, tumor differentiation, Child-Pugh class classification, and BCLC stage. This further suggests that preoperative ILIS is closely related to the tumor burden and liver function status of HCC patients. It has been established in previous studies that the prognosis of HCC patients is related to tumor burden and is influenced by liver function and the general condition of the patient [23,24]. Through univariate analysis, we found that tumor size, degree of tumor differentiation, vascular cancer thrombus, BCLC stage, and preoperative ILIS were all important prognostic factors for OS and RFS in HCC patients. However, after multifactorial analysis, only ILIS and BCLC stage were independent risk factors for OS in HCC patients; only the BCLC stage was an independent risk factor for RFS in HCC patients. Kaplan-Meier survival analysis also showed that patients with higher preoperative ILIS had shorter OS and RFS.
Nomograms are also a useful method for predicting survival outcomes [25]. According to the column line graphs, factors such as tumor size, degree of differentiation, and ILIS scores should be closely monitored to improve the clinical management of patients. Furthermore, it was also predicted that each patient with early to mid-stage HCC can survive at 3 and 5 years postoperatively, which has not been possible using other conventional predictive models. Hence, our column line plots showed good calibration and discrimination. A recent technology, ANN, has also been used to explore other important factors in predicting clinical patient survival [26]. Our results showed that the importance of BCLC staging and ILIS score was proportionally higher than other risk factors. Among these factors, ILIS is second only to BCLC staging, showing that ILIS has a pivotal role in OS.
There are several limitations to this study. First, the retrospective nature of the study may have led to selection bias. Fortunately, most of the baseline characteristics of the patients in the 2 cohorts were not significantly different. Secondly, the median OS was shorter in the validation cohort than in the training cohort, which may be due to the higher proportion of patients in the validation cohort being in the middle stages of BCLC, resulting in a generally worse condition and higher tumor burden. Hence, these factor may affect the accuracy of the column line graphs. Third, we did not include the analysis of other survival outcomes, such as the relationship between ILIS and progression-free survival (PFS) or disease-free survival (DFS). Finally, this study did not include patients with advanced HCC, a crucial factor to consider since HCC differs from other cancers since patients in advanced stages may undergo surgery in addition to palliative care, depending on their condition [27]. Hence, the validity and accuracy of preoperative ILIS as a prognostic guide for advanced HCC patients undergoing surgery is yet to be explored.
Conclusions
In conclusion, our study suggests that preoperative ILIS is a significant predictor of prognosis in patients with early to mid-stage HCC. Our findings suggest that clinicians should focus on the BCLC stage of HCC patients when formulating treatment plans and consider these readily available serum biomarkers associated with prognosis to provide better personalized treatment for HCC patients. In addition, we have constructed columnar plots and an ANN to assess the prognosis of early to mid-stage HCC patients undergoing radical hepatectomy. The model in this study has been observed to predict the survival of HCC patients more accurately. Further multicenter and large data studies are needed to validate the performance of ILIS in a more convincing manner.
Figures
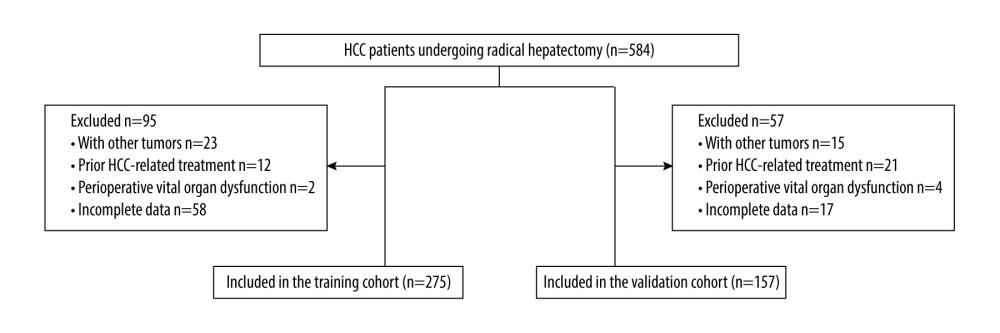 Figure 1. Patient selection process. HCC – hepatocellular carcinoma.
Figure 1. Patient selection process. HCC – hepatocellular carcinoma. 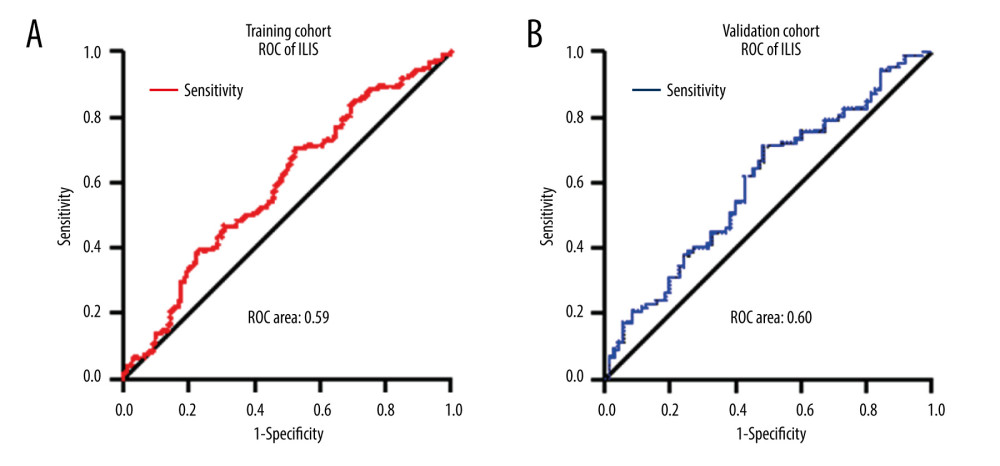 Figure 2. ILIS ROC curves of HCC patients. (A) The AUC of ILIS in the training cohort was 0.59. (B) The AUC of ILIS in the validation cohort was 0.60.
Figure 2. ILIS ROC curves of HCC patients. (A) The AUC of ILIS in the training cohort was 0.59. (B) The AUC of ILIS in the validation cohort was 0.60. 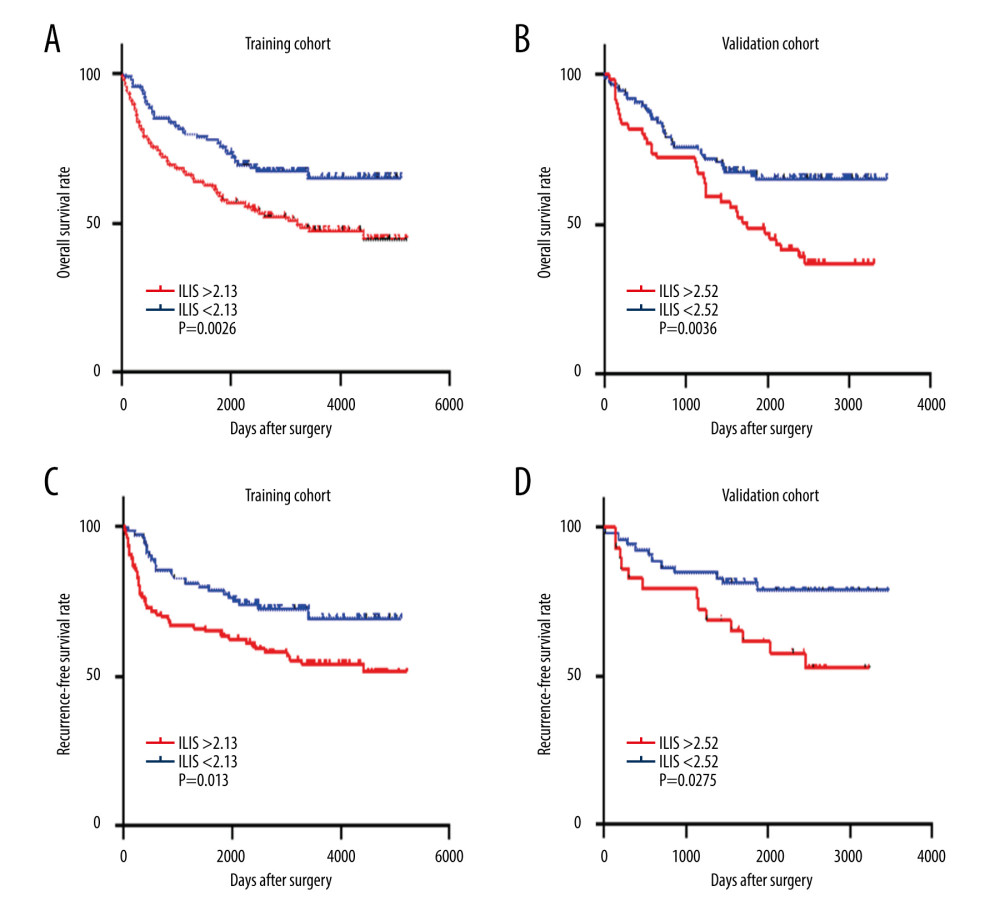 Figure 3. Kaplan-Meier survival analysis between the ILIS and OS/RFS of HCC patients after hepatectomy. Patients with higher ILIS score have a shorter OS (A, B) and RFS (C, D).
Figure 3. Kaplan-Meier survival analysis between the ILIS and OS/RFS of HCC patients after hepatectomy. Patients with higher ILIS score have a shorter OS (A, B) and RFS (C, D). 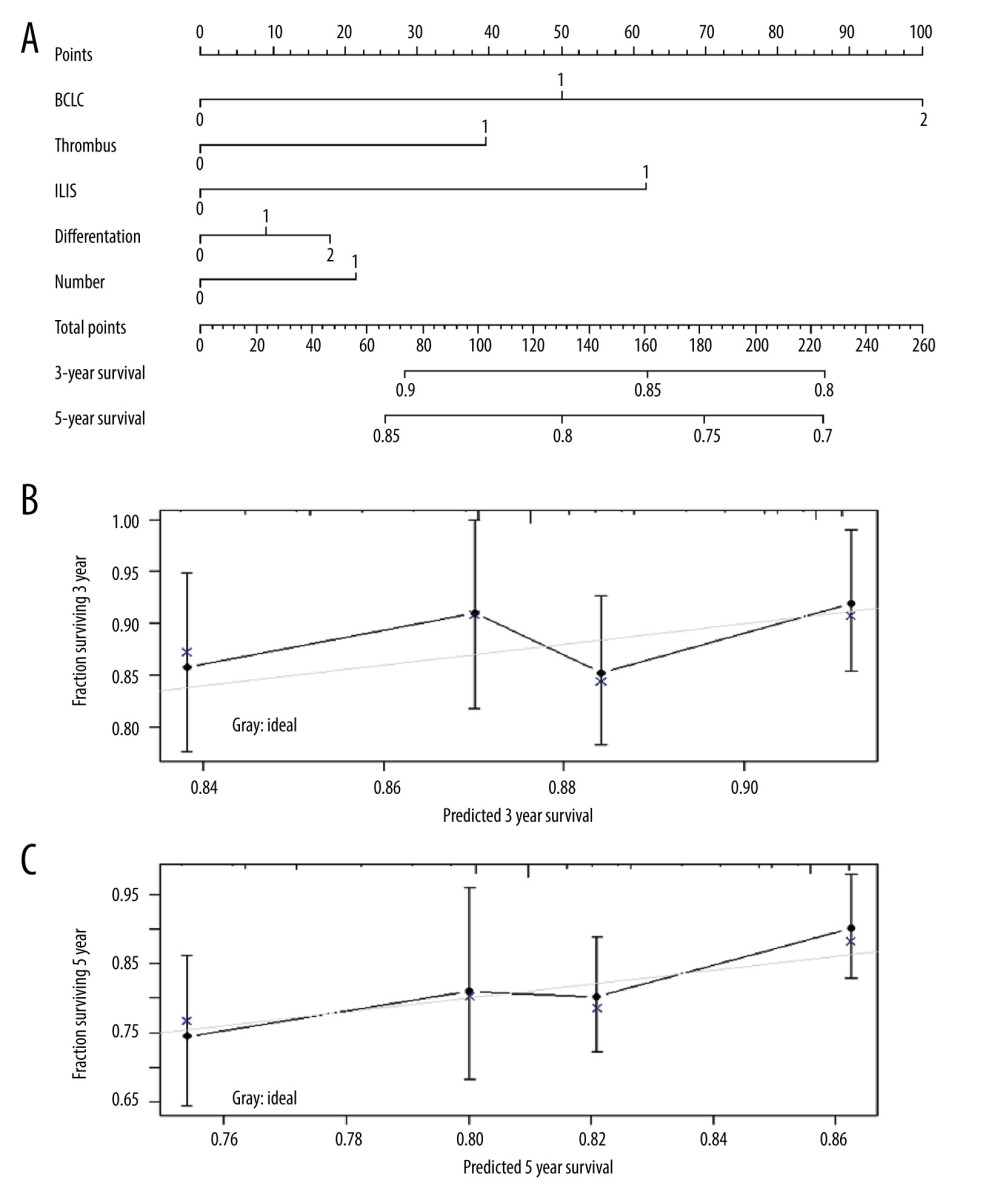 Figure 4. Nomogram predicting the probability of survival at 3 and 5 years post-operation. (A) ILIS-based nomogram for HCC patients after radical hepatectomy. (B, C) Good calibration for predicting survival at 3 and 5 years post operation.
Figure 4. Nomogram predicting the probability of survival at 3 and 5 years post-operation. (A) ILIS-based nomogram for HCC patients after radical hepatectomy. (B, C) Good calibration for predicting survival at 3 and 5 years post operation. 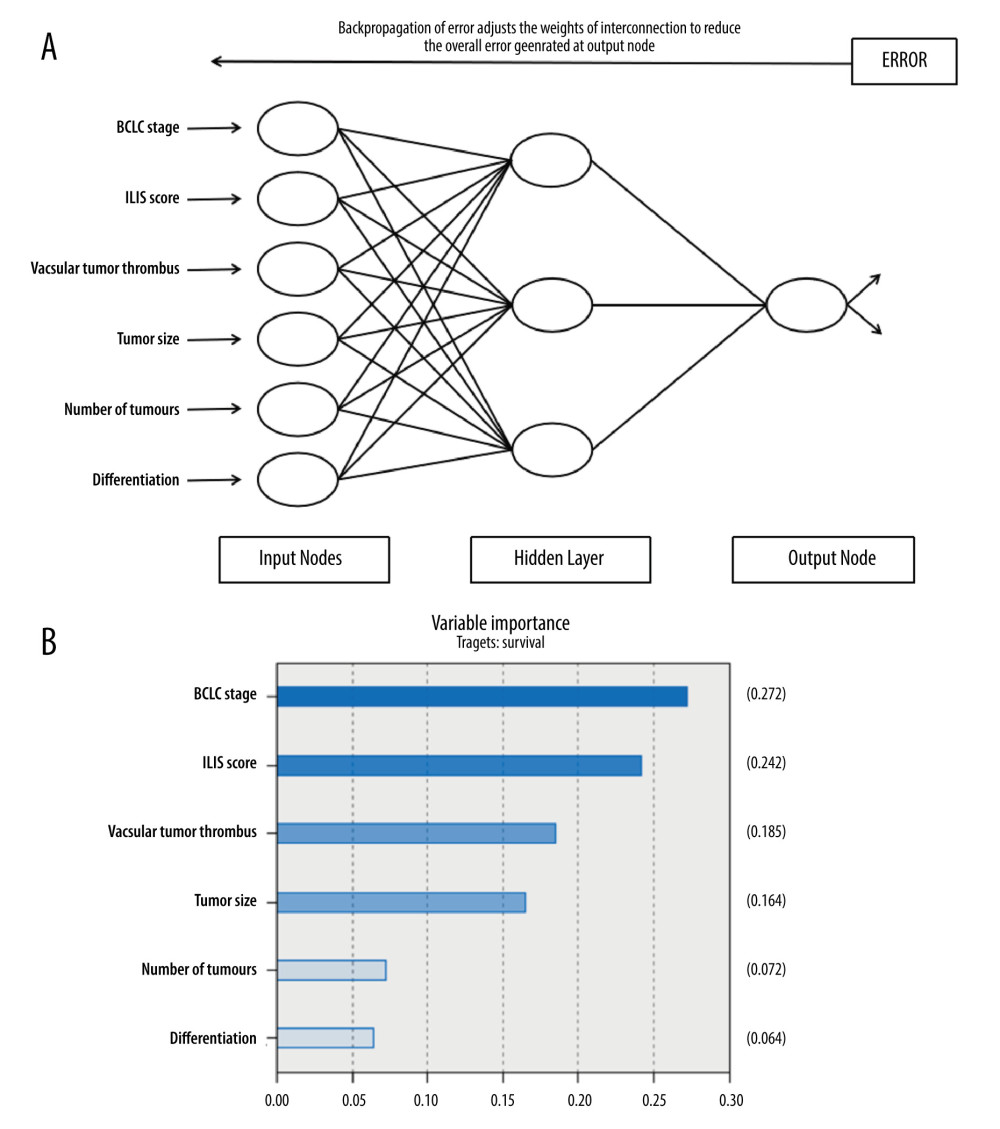 Figure 5. (A) Schematic representation of an ANN for predicting survival after radical hepatectomy in HCC patients. (B) Significance of the risk factors in the ANN model.
Figure 5. (A) Schematic representation of an ANN for predicting survival after radical hepatectomy in HCC patients. (B) Significance of the risk factors in the ANN model. References
1. Sung H, Ferlay J, Siegel RL, Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2021; 73(3); 209-49
2. Sun AW, Chne YS, Li LHepatitis B and hepatocellular carcinoma: Zhongguo Yi Miao He Mian Yi, 2010; 16(2); 173-77 [in Chinese]
3. Yi PS, Huang M, Zhang M, Comparison of transarterial chemoembolization combined with radiofrequency ablation therapy versus surgical resection for early hepatocellular carcinoma: Am Surg, 2018; 84(2); 282-88
4. Chen L, Guo P, He Y, CC-derived exosomes elicit HCC progression and recurrence by epithelial-mesenchymal transition through MAPK/ERK signalling pathway: Cell Death Dis, 2018; 9(5); 513
5. Tsai YC, Sou FM, Liu YW, Preoperative ALBI grade predicts the outcomes in non-B non-C HCC patients undergoing primary curative resection: BMC Gastroenterol, 2021; 21(1); 386
6. Shindoh J, Kawamura Y, Kobayashi Y, Platelet-albumin score as a sensitive measure for surgical risk prediction and survival outcomes of patients with hepatocellular carcinoma: J Gastrointest Surg, 2019; 23(1); 76-83
7. Kokudo T, Hasegawa K, Amikura K, Assessment of preoperative liver function in patients with hepatocellular carcinoma – the Albumin-Indocyanine Green Evaluation (ALICE) grade: PLoS One, 2016; 11(7); e0159530
8. Chan SL, Wong LL, Chan KA, Development of a novel inflammation-based index for hepatocellular carcinoma: Liver Cancer, 2020; 9(2); 167-81
9. Yu MQ, An TZ, Li JX, Integrated Liver inflammatory score predicts the therapeutic outcome of patients with hepatocellular carcinoma after transarterial chemoembolization: J Vasc Interv Radiol, 2021; 32(8); 1194-202
10. Bruix J, Sherman MAmerican Association for the Study of Liver Disease, Management of hepatocellular carcinoma: An update: Hepatology, 2011; 53; 1020-22
11. European Association for the Study of the Liver, European Organisation For Research and Treatment of Cancer, EASL-EORTC clinical practice guidelines: Management of hepatocellular carcinoma: J Hepatol, 2012; 56; 908-43
12. Chan AW, Kumada T, Toyoda H, Integration of albumin-bilirubin (ALBI) score into Barcelona Clinic Liver Cancer (BCLC) system for hepatocellular carcinoma: J Gastroenterol Hepatol, 2016; 31(7); 1300-6
13. Cross SS, Harrison RF, Kennedy RL, Introduction to neural networks: Lancet, 1995; 346; 1075-79
14. Duffy JP, Hiatt JR, Busuttil RW, Surgical resection of hepatocellular carcinoma: Cancer J, 2008; 14(2); 100-10
15. Nakagawa S, Hayashi H, Nitta H, Scoring system based on tumor markers and Child-Pugh classification for HCC patients who underwent liver resection: Anticancer Res, 2015; 35(4); 2157-63
16. Griffioen AW, Molema G, Angiogenesis: Potentials for pharmacologic intervention in the treatment of cancer, cardiovascular diseases, and chronic inflammation: Pharmacol Rev, 2000; 52(2); 237-68
17. Chiba T, Marusawa H, Ushijima T, Inflammation-associated cancer development in digestive organs: Mechanisms and roles for genetic and epigenetic modulation: Gastroenterology, 2012; 143(3); 550-63
18. Wang M, Wang Y, Feng X, Contribution of hepatitis B virus and hepatitis C virus to liver cancer in China north areas: Experience of the Chinese National Cancer Center: Int J Infect Dis, 2017; 65; 15-21
19. Meira JD, Fonseca GM, Carvalho Neto FN, Platelet-albumin (PAL) score as a predictor of perioperative outcomes and survival in patients with hepatocellular carcinoma undergoing liver resection in a Western center: Surg Oncol, 2022; 42; 101752
20. Young LB, Tabrizian P, Sung J, Survival analysis using albumin-bilirubin (ALBI) grade for patients treated with drug-eluting embolic transarterial chemoembolization for hepatocellular carcinoma: J Vasc Interv Radiol, 2022; 33(5); 510-517e1
21. Wong GL, Chan HL, Tse YK, On-treatment alpha-fetoprotein is a specific tumor marker for hepatocellular carcinoma in patients with chronic hepatitis B receiving entecavir: Hepatology, 2014; 59(3); 986-95
22. Ding Y, Liu K, Xu Y, Combination of inflammatory score/liver function and AFP improves the diagnostic accuracy of HBV-related hepatocellular carcinoma: Cancer Med, 2020; 9(9); 3057-69
23. Ho SY, Liu PH, Hsu CY, Tumor burden score as a new prognostic marker for patients with hepatocellular carcinoma undergoing transarterial chemoembolization: J Gastroenterol Hepatol, 2021; 36(11); 3196-203
24. Anger F, Klein I, Lv S, Preoperative liver function guiding HCC resection in normal and cirrhotic liver: Visc Med, 2021; 37(2); 94-101
25. Shen J, He L, Li C, Nomograms to predict the individual survival of patients with solitary hepatocellular carcinoma after hepatectomy: Gut Liver, 2017; 11(5); 684-92
26. Tetko IV, Neural network studies. 4. Introduction to associative neural networks: J Chem Inf Comput Sci, 2002; 42(3); 717-28
27. Zhu J, Yin T, Xu Y, Therapeutics for advanced hepatocellular carcinoma: Recent advances, current dilemma, and future directions: J Cell Physiol, 2019; 234(8); 12122-32
Figures
 Figure 1. Patient selection process. HCC – hepatocellular carcinoma.
Figure 1. Patient selection process. HCC – hepatocellular carcinoma. Figure 2. ILIS ROC curves of HCC patients. (A) The AUC of ILIS in the training cohort was 0.59. (B) The AUC of ILIS in the validation cohort was 0.60.
Figure 2. ILIS ROC curves of HCC patients. (A) The AUC of ILIS in the training cohort was 0.59. (B) The AUC of ILIS in the validation cohort was 0.60. Figure 3. Kaplan-Meier survival analysis between the ILIS and OS/RFS of HCC patients after hepatectomy. Patients with higher ILIS score have a shorter OS (A, B) and RFS (C, D).
Figure 3. Kaplan-Meier survival analysis between the ILIS and OS/RFS of HCC patients after hepatectomy. Patients with higher ILIS score have a shorter OS (A, B) and RFS (C, D). Figure 4. Nomogram predicting the probability of survival at 3 and 5 years post-operation. (A) ILIS-based nomogram for HCC patients after radical hepatectomy. (B, C) Good calibration for predicting survival at 3 and 5 years post operation.
Figure 4. Nomogram predicting the probability of survival at 3 and 5 years post-operation. (A) ILIS-based nomogram for HCC patients after radical hepatectomy. (B, C) Good calibration for predicting survival at 3 and 5 years post operation. Figure 5. (A) Schematic representation of an ANN for predicting survival after radical hepatectomy in HCC patients. (B) Significance of the risk factors in the ANN model.
Figure 5. (A) Schematic representation of an ANN for predicting survival after radical hepatectomy in HCC patients. (B) Significance of the risk factors in the ANN model. Tables
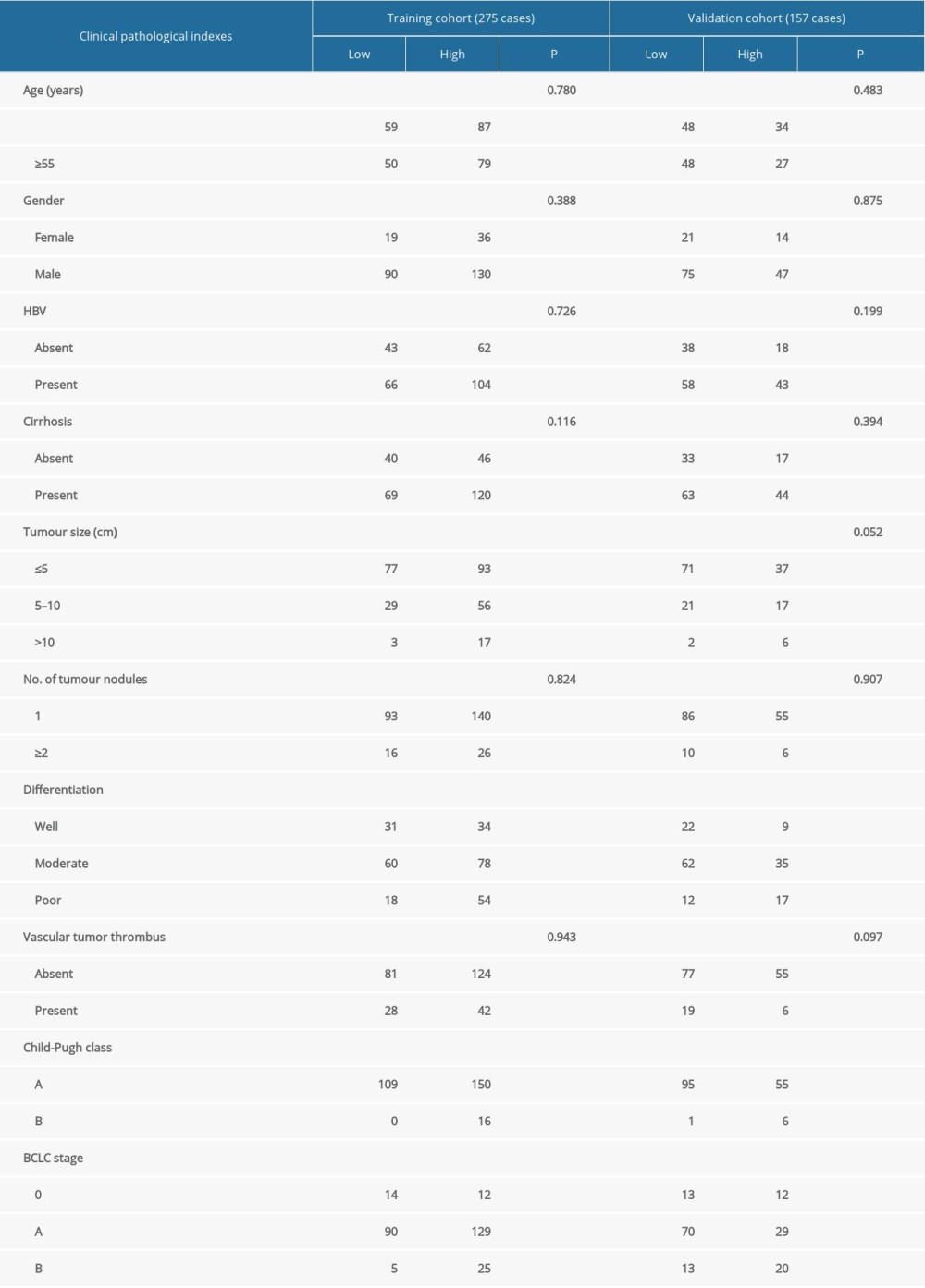 Table 1. Correlation of ILIS with clinicopathological characteristics.
Table 1. Correlation of ILIS with clinicopathological characteristics.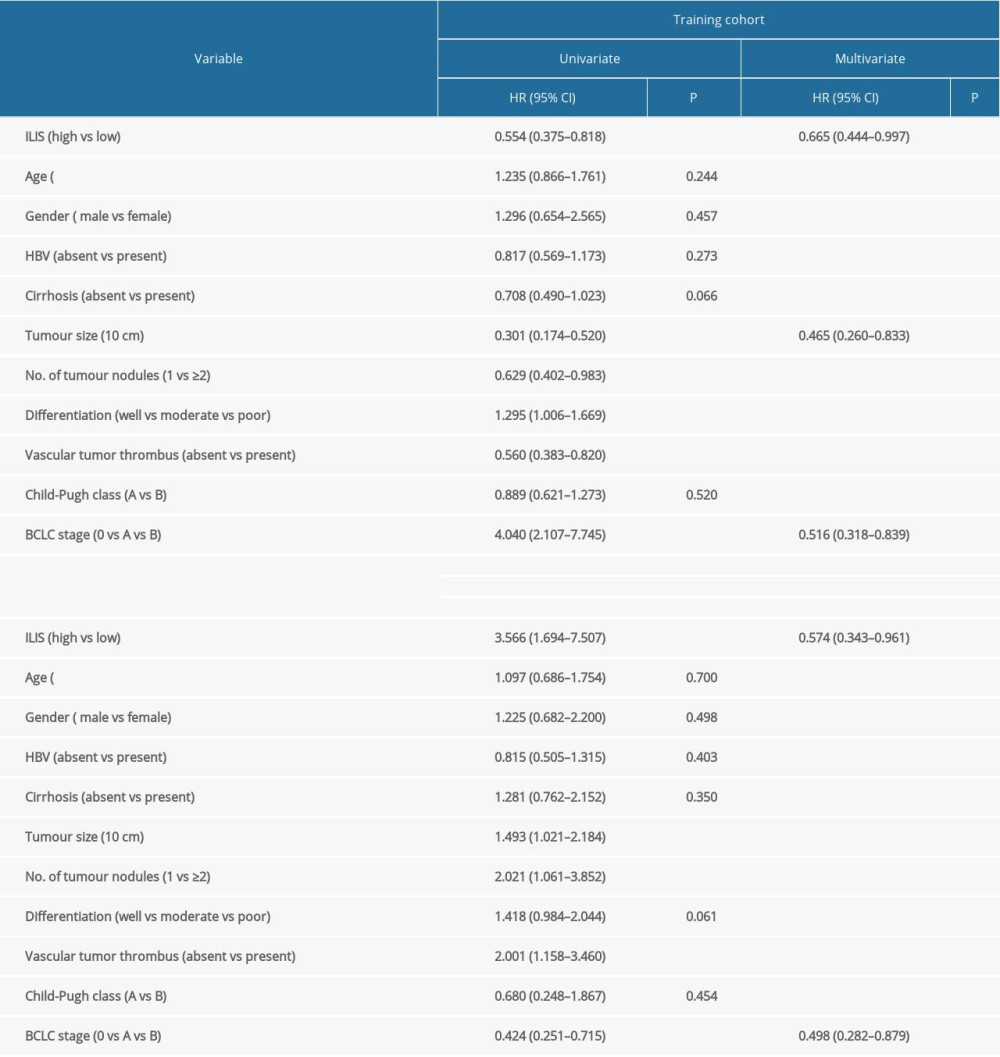 Table 2. Univariate and multivariate analyses of prognostic factors with OS in patients with HCC.
Table 2. Univariate and multivariate analyses of prognostic factors with OS in patients with HCC.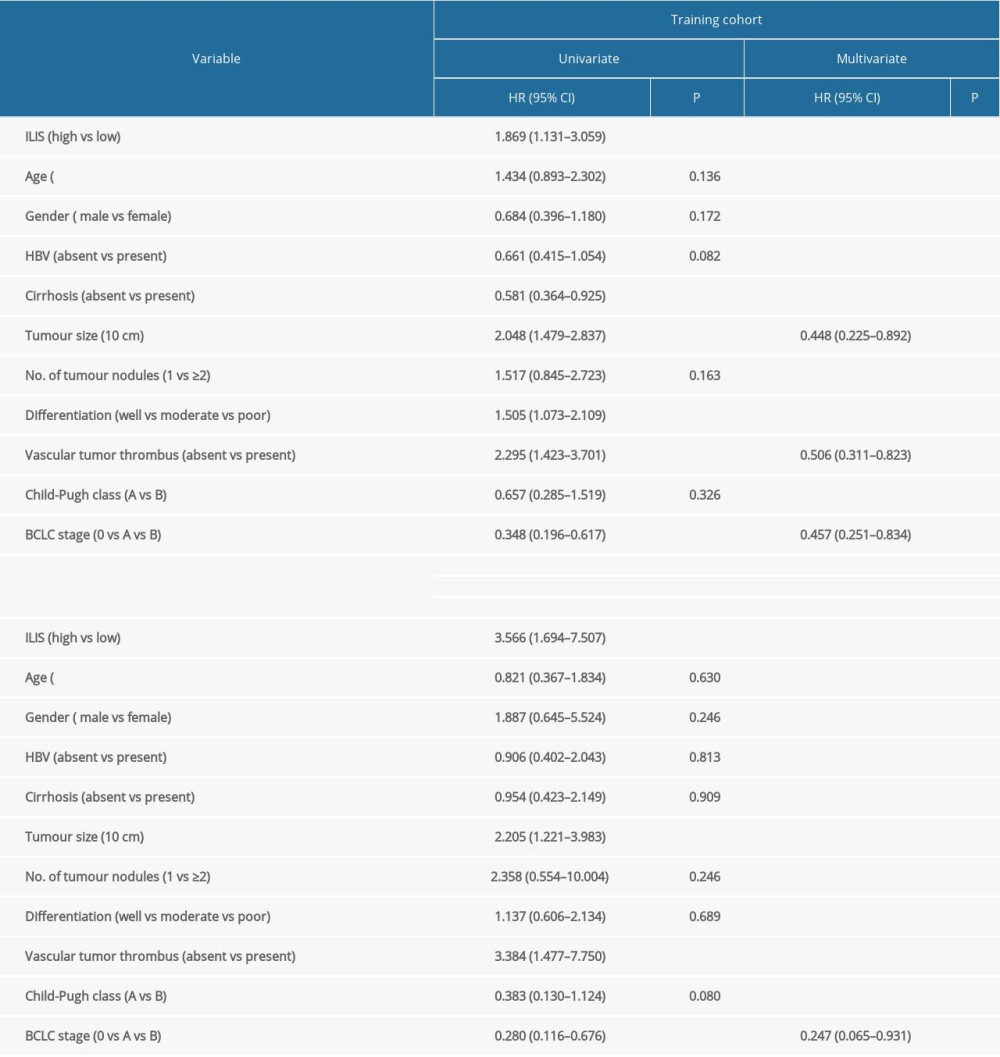 Table 3. Univariate and multivariate analyses of prognostic factors with RFS in patients with HCC.
Table 3. Univariate and multivariate analyses of prognostic factors with RFS in patients with HCC. Table 1. Correlation of ILIS with clinicopathological characteristics.
Table 1. Correlation of ILIS with clinicopathological characteristics. Table 2. Univariate and multivariate analyses of prognostic factors with OS in patients with HCC.
Table 2. Univariate and multivariate analyses of prognostic factors with OS in patients with HCC. Table 3. Univariate and multivariate analyses of prognostic factors with RFS in patients with HCC.
Table 3. Univariate and multivariate analyses of prognostic factors with RFS in patients with HCC. In Press
05 Mar 2024 : Clinical Research
Muscular Function Recovery from General Anesthesia in 132 Patients Undergoing Surgery with Acceleromyograph...Med Sci Monit In Press; DOI: 10.12659/MSM.942780
05 Mar 2024 : Clinical Research
Effects of Thermal Insulation on Recovery and Comfort of Patients Undergoing Holmium Laser LithotripsyMed Sci Monit In Press; DOI: 10.12659/MSM.942836
05 Mar 2024 : Clinical Research
Role of Critical Shoulder Angle in Degenerative Type Rotator Cuff Tears: A Turkish Cohort StudyMed Sci Monit In Press; DOI: 10.12659/MSM.943703
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








