27 January 2023: Clinical Research
Impact of Perioperatively Administered Amino Acids, Lidocaine, and Magnesium on Inflammatory Response and Pain Associated with Abdominal Surgery: A Prospective Clinical Study
Ognjenka Sarenac12ABDEF, Ranko Lazovic23BDEF, Batric Vukcevic32BCE*, Jelena R. Lazovic4BCDE, Ivan G. Palibrk5ADEFDOI: 10.12659/MSM.939204
Med Sci Monit 2023; 29:e939204
Abstract
BACKGROUND: Abdominal surgery is associated with a systemic inflammatory response which facilitates postoperative complications through immune imbalance and hypercatabolism. The aim of the study was to evaluate the effect of the combined perioperative lidocaine, magnesium, and amino acids on postoperative inflammation and pain.
MATERIAL AND METHODS: This prospective, randomized, double-blind study included 2 groups of patients undergoing abdominal surgery: Group 1 – receiving the aforementioned substances; and Group 2 – control (undergoing conventional general anesthesia). The following parameters were evaluated intraoperatively: arterial blood pressure, end-tidal CO₂ level, urine output, bispectral index, base excess, oxygen saturation, operating room temperature and body temperature (BT), opioid use, and surgery duration; and postoperatively: total leukocyte (WBC), neutrophil, lymphocyte and platelet count; fibrinogen, C-reactive protein (CRP), procalcitonin (PCT) and interleukin (IL)-6 levels; numeric rating scale (NRS) pain level, first flatus and bowel movement, and postoperative complications. The postoperative parameters were evaluated 2 h and 6 h postoperatively, as well as on postoperative days (POD) 1, 2, 3, and 5.
RESULTS: Group 1 showed lower counts of WBC, neutrophils, and lymphocytes and lower fibrinogen, CRP, PCT, IL-6, and BT in the first 5 POD, as well as NRS scores and time to first flatus/bowel movement. The groups did not differ significantly regarding postoperative complications.
CONCLUSIONS: The isolated effects of lidocaine, magnesium, and amino acids in surgery have been described previously. To the best of our knowledge, this is the first study to show the anti-inflammatory and analgesic effects of simultaneous use of these substances in abdominal surgery.
Keywords: Amino Acids, general surgery, Lidocaine, Magnesium, Humans, Prospective Studies, Flatulence, Pain, Postoperative, Analgesics, Opioid, Postoperative Complications, Double-Blind Method
Background
Surgery is associated with local tissue damage and alterations in the physiological mechanisms of homeostasis [1,2]. SIRS (systemic inflammatory response syndrome) occurs in 16% to 89% of abdominal surgical patients [3–5]. IL-6 (interleukin-6) is the key proinflammatory cytokine, responsible for the activation of acute phase reactants CRP (C-reactive protein) and PCT (procalcitonin) [5]. PCT [6] and IL-6 [1,7,8] levels are directly correlated with the intensity of surgical trauma.
Postoperative pain involves a combination of inflammatory and neuropathic mechanisms, both associated with hyperalgesia. The imperative of fast-track protocols is adequate analgesia with few adverse effects, enabling the patient to function independently and to be discharged sooner [9]. Effective multimodal analgesia which reduces the need for opioid drugs is one of the key elements in ERAS (enhanced recovery after surgery) protocols [10].
Lidocaine, a local anesthetic drug, blocks the sodium-potassium channel [11], thus alleviating postoperative pain [12–16] through the following mechanisms: analgesic, anti-hyperalgesic, and anti-nociceptive [17]. Furthermore, lidocaine serves as a N-methyl-D-aspartate (NMDA) receptor antagonist [18]. Perioperatively administered lidocaine also has anti-inflammatory activity [17,19–24] and facilitates the return of bowel function after abdominal surgery [9,16,23,25].
Perioperative amino acid solutions have a positive thermogenic effect [26–28] and anti-inflammatory activity [29–31] in surgical patients.
Magnesium is a NMDA receptor antagonist (as is lidocaine) [32], central nervous system depressor (through the effect on calcium channels), and sodium-potassium gate modulator [33]. There is evidence of its analgesic [32–36] and anti-inflammatory effect [37,38] in surgical patients.
The reduced postoperative systemic inflammatory response decreases the risk of postoperative complications through alleviation of immune system dysregulation and hypercatabolism. The aforementioned studies most commonly described the separate effects of lidocaine, magnesium, and amino acids on postoperative inflammatory response or pain. The aim of the present study was to examine the influence of simultaneously administered amino acids, lidocaine, and magnesium on postoperative inflammatory response and pain in abdominal surgery.
Material and Methods
ETHICS STATEMENT:
This prospective, double-blind study was conducted from September 2021 to August 2022 on 100 patients (divided into 2 groups of 50 patients each) treated in the Clinical Center of Montenegro (Podgorica, Montenegro). The study was approved by the Ethics Committee of the Clinical Center of Montenegro (03/01-6031/1). The research was conducted in accordance with the Declaration of Helsinki. Every patient signed the written informed consent form before inclusion.
INCLUSION CRITERIA:
This prospective, randomized, double-blind study included 2 patient groups: Group 1 was the experimental group (receiving amino acids, lidocaine, and magnesium perioperatively, along with the conventional anesthesia procedures) and Group 2 was the control group (undergoing conventional anesthesia). The research was conducted on patients of both sexes, age 35–75 years, undergoing 1 of the following surgical procedures in general anesthesia: total/subtotal gastrectomy, small bowel resection and anastomosis, right/extended right/left/subtotal/total colectomy and anastomosis; or high anterior/low anterior resection.
THE ANESTHESIA PROTOCOL:
All patients were preoperatively examined and prepared for surgery by an anesthesiologist according to the comorbidities and the planned intervention according to the local protocol. The patients were rehydrated during the day before surgery by drinking water or tea, as well as intravenous 1000 ml Ringer-lactate and 1000 ml 5% dextrose solution with or without 6 international units of rapid-acting insulin (adjusted according to glycemia levels in diabetic patients). Solid foods were not consumed within 24 h before surgery and liquids (including high-calorie liquid foods) were not consumed within 4 h before surgery. The patients were administered 1 mg oral alprazolam 6 h before surgery and 40 mg i.v. pantoprazole 1 h before surgery. The antibiotic prophylaxis with first-generation cephalosporin and metronidazole was conducted 60 min before the incision was made. In case of antibiotic allergy, 600 mg i.v. clindamycin and 240 mg i.v. gentamycin were used. All patients received low-molecular-weight heparin 12 h before surgery (dose adjusted to body mass and comorbidities).
The patients in both groups underwent intravenous anesthesia induction and maintenance, with previous use of 100% oxygen. Propofol 2–2.5 mg/kg was administered, as well as 5 mcg/kg fentanyl and 0.2 mcg/kg cisatracurium. Tracheal intubation was performed using an endotracheal tube with cuff. A nasogastric tube and a central venous catheter were inserted. Invasive arterial pressure monitoring was used. Body temperature (BT) was measured using a nasopharyngeal temperature probe. Hourly urine output was monitored. A Primus® (Draeger) ventilator was used. Mechanical ventilation was pressure-balanced (5 mmHg positive end-expiratory pressure (PEEP), 7 mmHg pressure support, 20 mmHg peak inspiratory pressure (PIP), and breath rate 10–12/min). End-tidal CO2 (EtCO2) levels were in the normocapnia range (30–45 mmHg). Anesthesia maintenance was achieved using hypnotic volume concentrations of sevoflurane (0.7%), while the anesthesia was opioid-based. Muscle relaxation was maintained with bolus infusions of cisatracurium. We administered 7–10 mg intravenous morphine approximately 40 min before the end of surgery. At the end of the procedure, the neuromuscular blockade was reversed (using 2.5 mg neostigmine and 1 mg atropine. Bispectral index (BIS) was maintained at 40–60.
During surgery, fluid and electrolyte resuscitation, as well as blood transfusion and blood products, were administered based on the full blood count, coagulation factor levels and clotting time, hemodynamic and EtCO2 monitoring changes, urine output, and the occurrence of intestinal edema and conjunctival suffusion. Also, visible blood loss was monitored, and the insensible fluid losses were estimated. All intravenous infusions were heated to 37°C. Patients were covered with regular sterile blankets (without heating) during surgery.
All patients received the same postoperative analgesic and antiemetic therapy (24-h i.v. infusion of 300 mg tramadol, 5 mg metamizole-sodium, and 3 mg ondansetron in 500 ml 0.9% saline, administered at an average rate of 25 ml/h). Based on the clinical signs and the patient’s subjective perception, an additional bolus of 2 mg i.v. morphine was used in case of pain degree > 5 on the numeric rating scale (NRS). CPAP (continuous positive airway pressure) ventilation was used during the first 2 postoperative hours in all patients to prevent lung atelectasis. Verticalization was initiated 6–8 h postoperatively.
RANDOMIZATION AND STUDY PROTOCOL:
We prepared 50 cards marked “Group 1” and 50 cards marked “Group 2” before the beginning of the study. Before the procedure start time, the researcher pulled a card for the specific patient and gave it to the anesthetist in the preoperative unit, who then prepared the appropriate solutions.
Based on the card pulled for the specific patient, the anesthetist in the preoperative unit prepared the solutions and gave them to the anesthetist in the operating room (OR). Thus, the researcher, as well as the anesthesiologist and the anesthetist treating the patient in the OR, did not know which group the patient was in. In the event of allergy and other unexpected events, the group was revealed and the patient was excluded from the study.
Patients in Group 1 received additional i.v. 2% lidocaine: 100 mg diluted in 100 ml 0.9% saline infused over 30 min before the incision (at a rate of 200 ml/h). This was followed by 300 mg i.v. lidocaine diluted in 300 ml 0.9% saline administered during the procedure at a rate of 250 ml/h, and this was discontinued after infusion termination. The maximum lidocaine dose did not exceed 300 mg/h. Also, Group 1 received i.v. 1% magnesium-sulphate (2 g diluted in 100 ml 0.9% saline infused over 20 min before the incision); as well as i.v. 10% amino acid solution Aminosol® (manufactured by Hemomont, Podgorica, Montenegro) administered after the induction of anesthesia as 500-ml infusions at a maximum rate of 250 ml/h until the end of surgery (the maximum dose being 2 g/kg). Aminosol® contains the following amino acids: isoleucine, leucine, valine, lysine, methionine, threonine, phenylalanine, alanine, arginine, glycine, histidine, proline, serine, tyrosine, taurine, and tryptophan [39].
The patients in Group 2 received 100 ml 0.9% saline administered 30 min before incision (at a rate of 200 ml/h), 300 ml 0.9% saline administered during surgery at a rate of 250 ml/h, 100 ml 0.9% saline infused over 20 min before the incision, 500 ml 0.9% saline administered during surgery and after induction of anesthesia as a 500-ml infusion at a maximum rate of 250 ml/h until the end of surgery, as per the regimen used in Group 1. All intravenous fluid containers were marked such that the anesthesiologist and the anesthetist in the OR were unaware of their contents.
The following parameters were measured:
The aforementioned postoperative parameters were measured 6 times for each patient:
Furthermore, the POD of the first postoperative flatus or bowel movement was registered for each patient, as well as the occurrence of postoperative surgical complications according to the Clavien-Dindo classification [40].
STATISTICAL ANALYSIS:
Statistical analysis was performed using descriptive and analytical statistics. Categorical variables were compared using the chi-squared test. The normality of distribution was assessed using the Kolmogorov-Smirnov test. Continuous variables not normally distributed were compared using the Mann-Whitney U test. As an additional analysis, the receiver-operating characteristic (ROC) curve with the calculation of the area under the curve (AUC) was used to indirectly assess the possible association of amino acids, lidocaine, and magnesium use with the levels of the postoperative inflammatory and pain parameters. SPSS software for Windows 22.0 was used.
Results
PREOPERATIVE DATA:
The groups did not differ significantly by sex, age, indications for surgery, or other preoperative parameters excluding the PLT count (higher in Group 1) and IL-6 level (lower in Group 1). The most common indication for surgery in both groups was rectal cancer (Table 1).
Arterial hypertension was the most common comorbidity in both groups. The groups did not differ significantly by comorbidities (Table 2).
INTRAOPERATIVE DATA:
Intraoperative data are presented in Table 3. EtCO2 and base excess were significantly different between Groups 1 and 2, as well as the BTB and TORA–C.
Intraoperative opioid usage was not different between Groups 1 and 2 (969.00±222.67 vs. 915.00±163.90,
POSTOPERATIVE DATA:
Postoperative variations of cellular counts; fibrinogen, CRP, PCT, and IL-6 levels, as well as axillary BT and NRS scores, are presented in Figures 1–3.
First flatus usually occurred on POD 2 (2.12±0.59 days) in Group 1 and POD 4 in Group 2 (4.16±1.02 days). First stool occurred usually on POD 3 (3.18±0.80 days) and POD 6 (6.40±1.29 days) in Groups 1 and 2, respectively. Group 1 showed significantly shorter times to first flatus and stool (
ROC curves were used to analyze and compare the AUC of postoperative parameters between Groups 1 and 2, which confirmed the differences listed in Figures 1–3. The AUC values are given in Table 4. Table 5 contains the postoperative complications in Groups 1 and 2 (no significant difference was found regarding the complication occurrence nor the Clavien-Dindo grades).
Discussion
Postoperative pain and ileus are regarded as the most important factors in treatment costs, due to the prolonged hospital stay [9]. Nearly 40% of colorectal surgery patients suffer from postoperative intestinal paresis [10]. Also, a specific problem in abdominal surgery is the occurrence of septic complications, which must be recognized and separated from the postoperative SIRS occurring as an inflammatory response to surgical trauma [41]. Past studies have assessed the influence of inflammatory markers in the discrimination between the physiological inflammatory response and the inflammation accompanying septic complications [42–47].
The research presented herein showed the anti-inflammatory effect of the combination of lidocaine, amino acids, and magnesium in abdominal surgery through lower counts of WBC, neutrophils, and lymphocytes, as well as lower levels of fibrinogen, CRP, PCT, IL-6, and BT, in the first 5 PODs. Furthermore, postoperative pain scores were lower in Group 1. These differences were confirmed with direct comparisons and AUC scores. Intraoperatively, both groups were in the normocapnia range, but Group 1 showed lower deviations in base excess and lower EtCO2, suggesting better oxygenation and, indirectly, a lower production of lactates.
Group 1 showed significantly lower WBC and lymphocyte counts on PODs 1–5, while the neutrophil count was lower on PODs 2–5. Leukocytosis occurs in the first 24 h after surgery, based on neutrophilia. Subsequently, neutrophils are removed from the circulation by the liver, spleen, and bone marrow [2]. PLT counts were higher in Group 1 in all measurements (mirroring the preoperative values), but without statistical significance. Fibrinogen peaked on POD 2 in both groups, with significantly lower levels in Group 1 on PODs 1–5. CRP, PCT, and IL-6 levels were significantly lower in Group 1 in all measurements, peaking on POD 1 in both groups. Group 1 had a significantly lower level of IL-6 preoperatively, which might have influenced postoperative values. However, the reduction of other inflammatory parameters confirms the anti-inflammatory effect of the studied combination of substances. Research on abdominal surgery patients showed a beneficial anti-inflammatory effect of amino acids (mostly glutamine and arginine), leading to lower IL-6 [29,48,49], IL-1, IL-18, TNF (tumor-necrosis factor)-α [29], and CRP levels [30,50,51]. The anti-inflammatory effect of lidocaine is shown by the reduction of postoperative IL-6 and IL-8 [19,22,23], IL-1 [19], and CRP levels [52]. There is limited evidence of the anti-inflammatory effect of magnesium in vascular surgery (lowering the IL-6 level) [37] and in cardiac surgery (lowering the IL-6 and TNF-α levels) [38].
General anesthesia is associated with hypothermia due to a reduction in metabolic thermogenesis, as well as impaired central thermoregulation [53,54]. A systematic review by Aoki et al including 14 randomized clinical trials (RCT) with 626 patients showed a thermogenic effect of perioperative amino acids, as well as faster extubation time and shorter hospital stay [26]. Our study showed intraoperative normothermia in both groups, with the only statistical difference in BT 2, which was lower in Group 1. Therefore, the thermogenic effect of amino acids was not shown. Group 1 had significantly lower postoperative BT in all measurements, most likely due to reduced systemic inflammation.
A Cochrane review from 2015 (45 RCTs, 2802 patients, 25 papers on abdominal surgery, and 20 papers on other surgical areas) showed reduced pain levels and opioid usage (in the first 24 h after surgery) in the group of patients receiving intravenous perioperative lidocaine. This analgesic effect was not shown 48 h after surgery. Lidocaine also reduced the incidence of postoperative ileus (4.8% versus 13.9% in the control group) [9]. Lidocaine is thought to be more effective in abdominal surgery than in other surgeries [55–57], which is somewhat confirmed in the repeated Cochrane review from 2018 (68 RCTs, 4525 patients, 42 papers on abdominal surgery, and 46 papers on other surgical specialties) [58]. This review showed a lower level of evidence of the analgesic and prokinetic effect of lidocaine, perhaps due to a higher percentage of non-abdominal surgery papers included in the review. Many studies on lidocaine focused exclusively on pain in the first 24 h [12–14] or 48 h [15] after surgery. The results of studies examining the analgesic effect of lidocaine 72 h after surgery are conflicting [16,59].
Albrecht et al published a meta-analysis in 2013 (25 studies, 1461 patients) showing the effect of perioperative magnesium on reduction of postoperative pain and need for opioid analgesics 24 h after surgery [34], comparable to the meta-analysis by De Oliveira et al from the same year (20 RCTs, 1257 patients) [32]. In 2017, Kim et al compared the effects of lidocaine and magnesium on postoperative pain in 126 patients undergoing mastectomy, showing that the 2 drugs had comparable analgesic effects in the immediate postoperative recovery period, as well as 24 h after surgery [18]. A similar sample was used in a study by Mendonca et al in 2020, which analyzed the combination of lidocaine and magnesium. Their results showed that these 2 drugs combined had superior analgesic effect (in the first 24 h after surgery) compared with the isolated use of lidocaine or magnesium, as well as the control group. The authors suggest that the simultaneous inhibition of NMDA receptors is the foundation of the synergism of these 2 drugs, as well as the attenuation of the membrane potential (modulating the transport of sodium, potassium, and calcium) and their anti-inflammatory effect [33]. Saadawy et al also indicate that combined lidocaine and magnesium might have a prolonged analgesic effect via NMDA receptor inhibition, as well as the prevention of spinal and peripheral hypersensitivity [36].
In our study, amino acids, lidocaine, and magnesium showed an anti-inflammatory and analgesic effect in the first 5 PODs (the postoperative period examined in the study), as well as having a prokinetic effect. It seems that the synergism of these substances results in a prolonged beneficiary effect on postoperative inflammation and pain. Furthermore, it seems that this combination is useful in abdominal surgery. The choice to analyze the first 5 PODs was made in order to encompass the peak of the most commonly used postoperative inflammatory parameters, as well as the beginning of their return to baseline. Based on the literature search, we think that this is the first study to examine the effect of simultaneous use of these 3 substances in abdominal surgery.
This study has several limitations, which can be overcome with additional research. We did not analyze the isolated effects of lidocaine, magnesium, and amino acids – all patients in Group 1 received all the substances. Also, the sample was too small for advanced statistics. Future studies should be performed on larger samples including other surgical specialties and laparoscopic surgery, with the comparison of the effects of these individual substances.
Conclusions
In this paper, we showed the anti-inflammatory effect of simultaneous lidocaine, magnesium, and amino acids perioperatively use in abdominal surgery, as well as their beneficial effect on postoperative pain and peristalsis. Postoperative inflammation triggers pain through multiple mechanisms. Therefore, these results suggest that a reduction in inflammation consequently reduces postoperative pain. This study has certain implications for clinical practice and future research on multimodal analgesia and anti-inflammatory perioperative therapy.
Figures
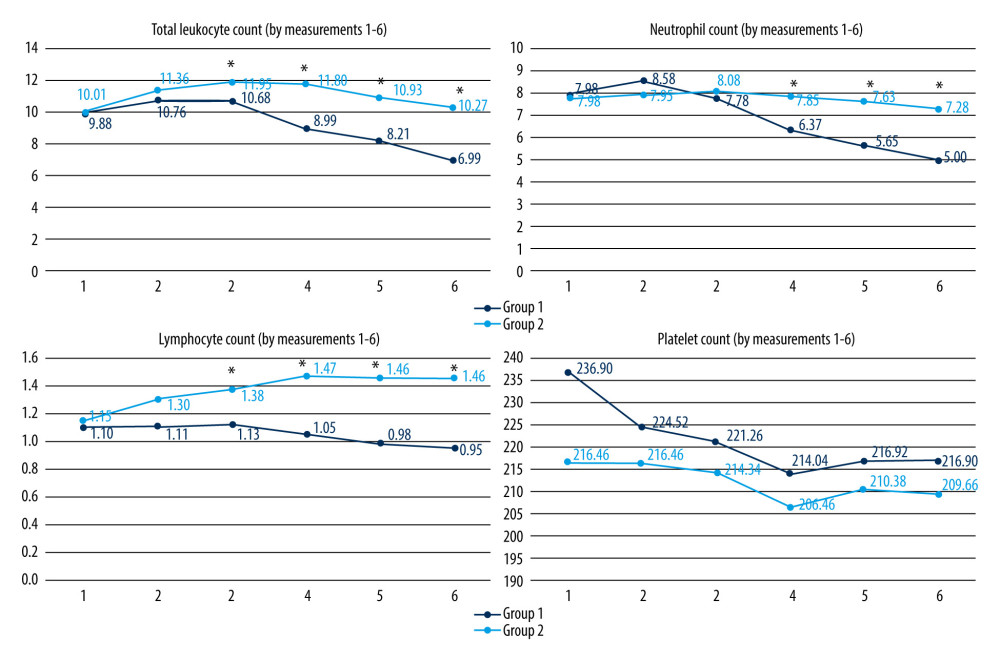 Figure 1. Postoperative WBC, neutrophil, lymphocyte, and PLT counts (109/l) (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test).
Figure 1. Postoperative WBC, neutrophil, lymphocyte, and PLT counts (109/l) (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test). 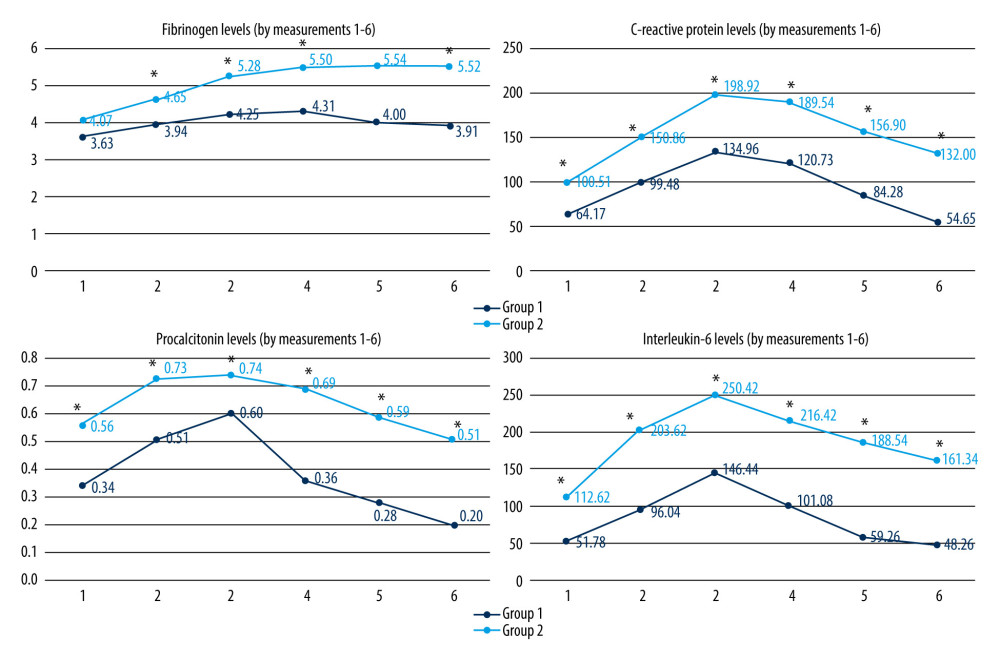 Figure 2. Postoperative levels of fibrinogen (g/l), CRP (mg/l), PCT (ng/ml) and IL-6 (pg/ml) (statistically significant difference is marked with an asterisk (P<0.001); Mann-Whitney U test).
Figure 2. Postoperative levels of fibrinogen (g/l), CRP (mg/l), PCT (ng/ml) and IL-6 (pg/ml) (statistically significant difference is marked with an asterisk (P<0.001); Mann-Whitney U test). 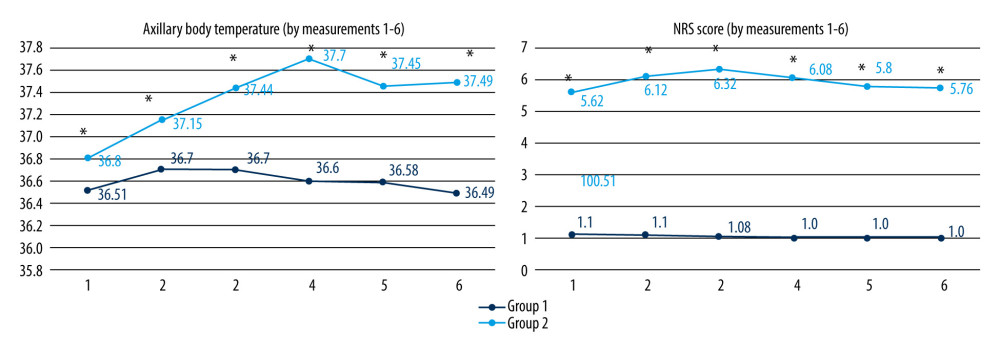 Figure 3. Postoperative axillary BT (°C) and NRS (numeric rating scale) scores (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test).
Figure 3. Postoperative axillary BT (°C) and NRS (numeric rating scale) scores (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test). Tables
Table 1. Preoperative parameters in Group 1 and Group 2 (statistically significant differences are in bold; P<0.05).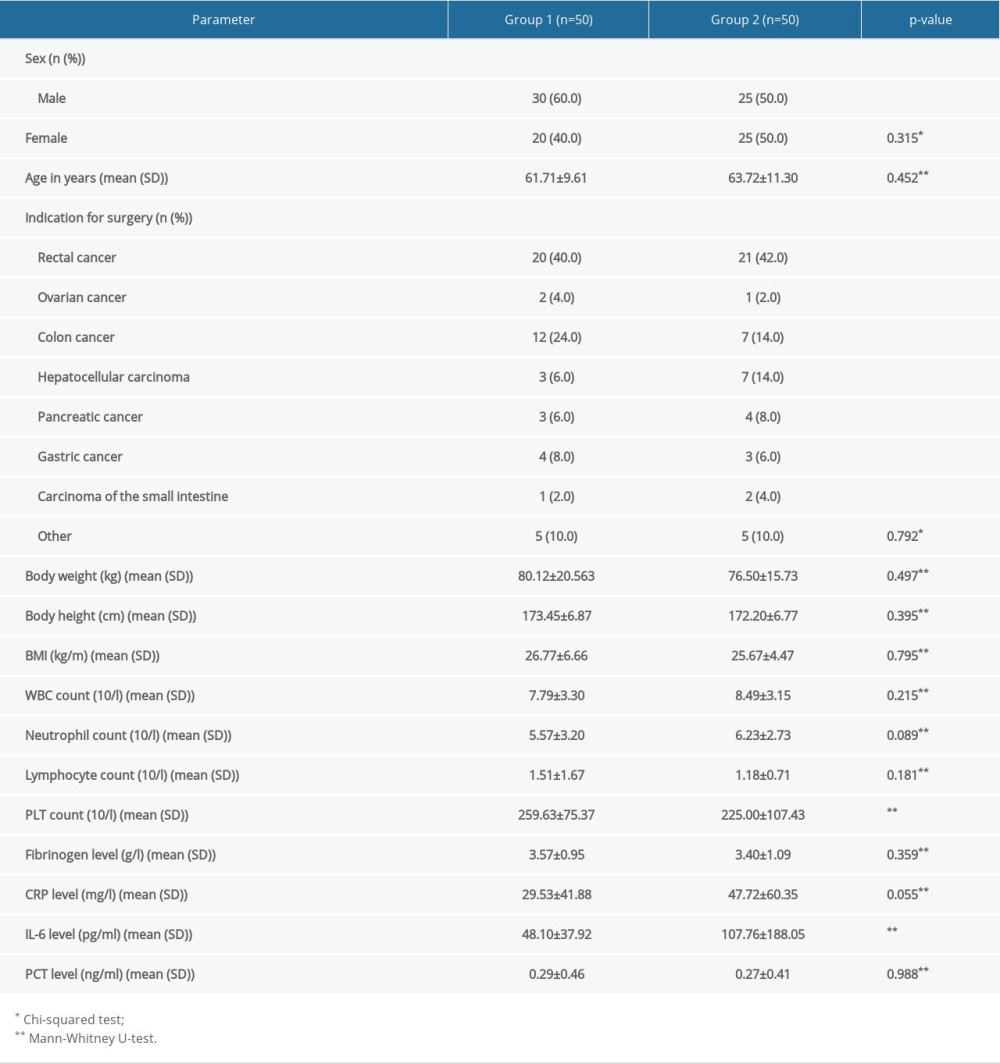 Table 2. Comorbidities in Group 1 and Group 2.
Table 2. Comorbidities in Group 1 and Group 2.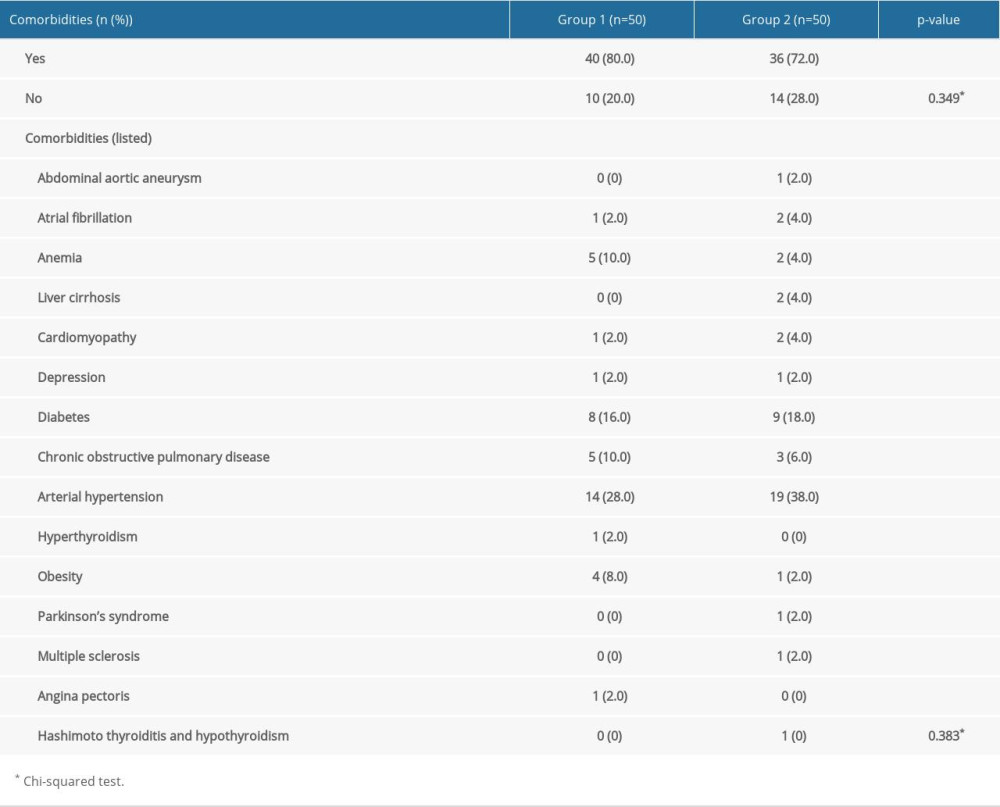 Table 3. Intraoperative parameters of Group 1 and 2 (statistically significant differences are in bold; P<0.05).
Table 3. Intraoperative parameters of Group 1 and 2 (statistically significant differences are in bold; P<0.05).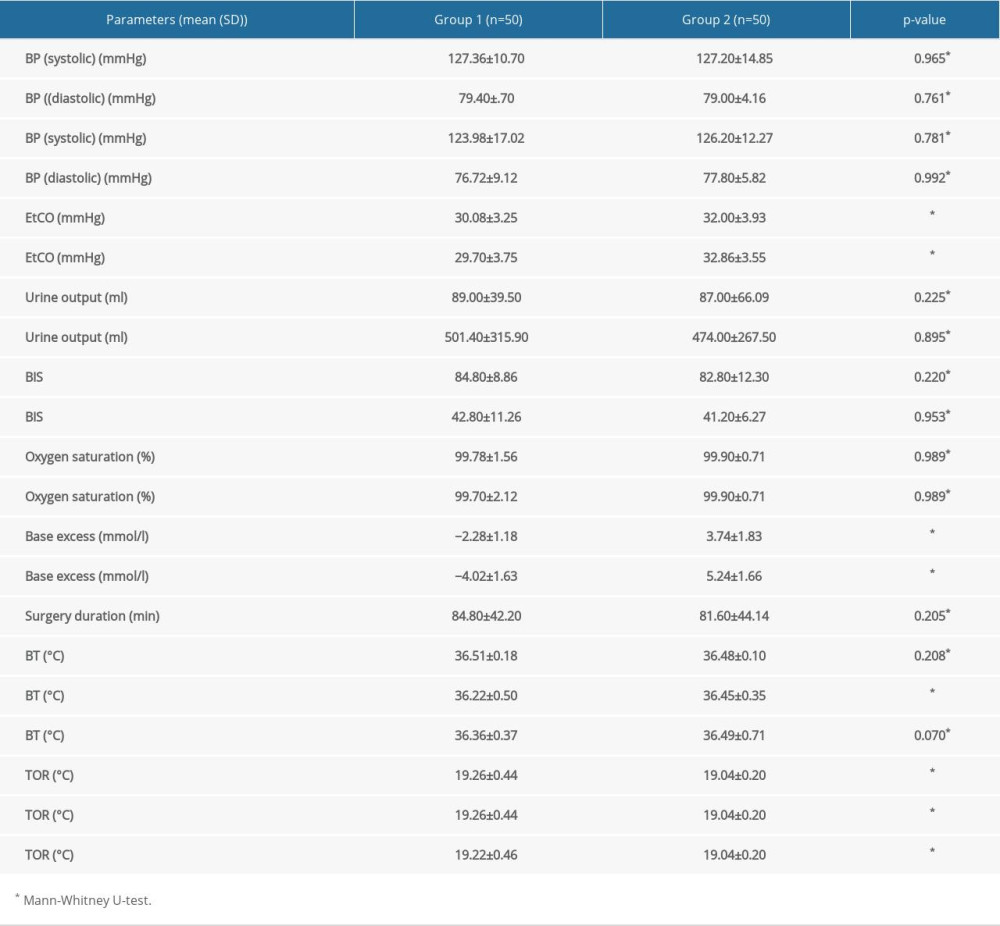 Table 4. ROC curve analysis and AUC calculation of the postoperative statistically significant parameters (excluding the PLT count) (statistically significant differences are in bold; P<0.05).
Table 4. ROC curve analysis and AUC calculation of the postoperative statistically significant parameters (excluding the PLT count) (statistically significant differences are in bold; P<0.05).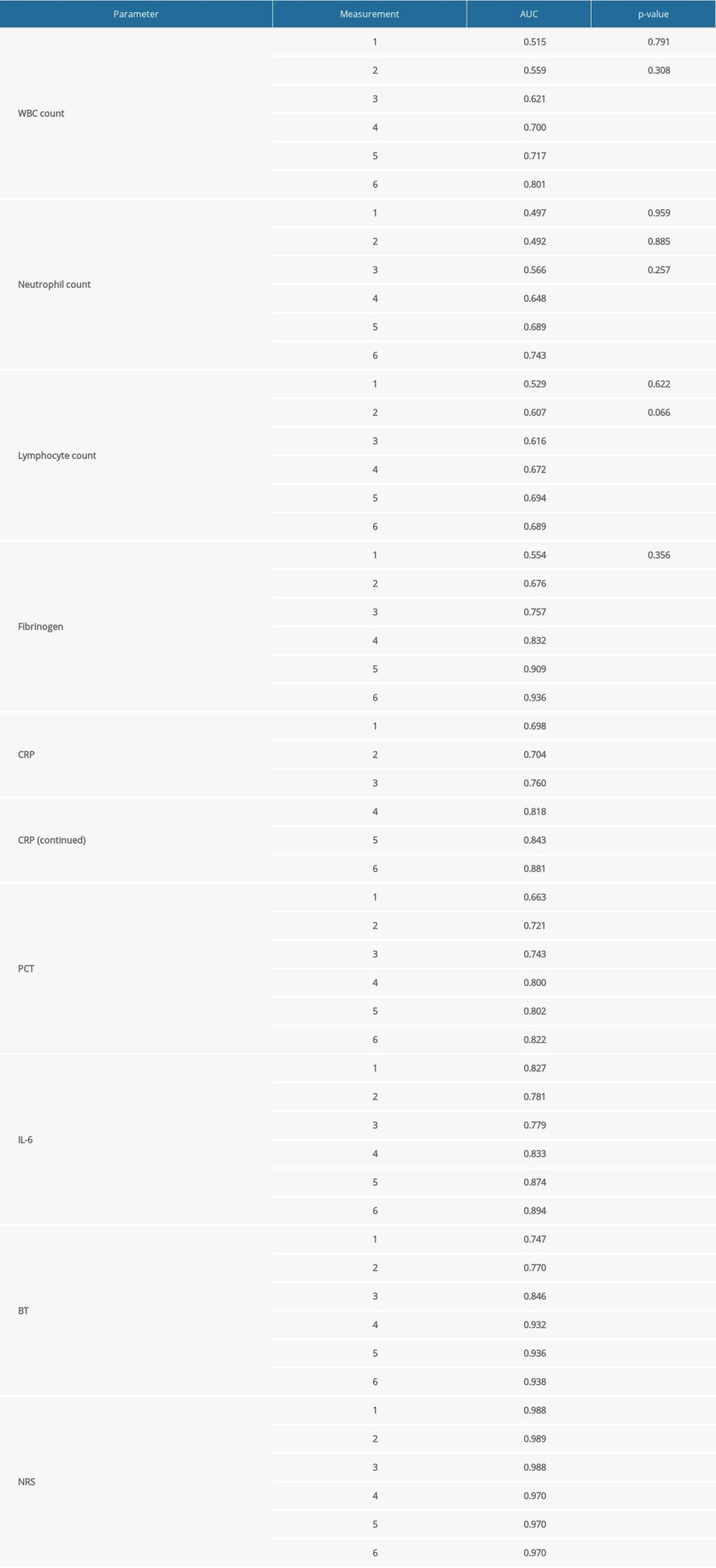 Table 5. Incidence of postoperative complications and Clavien-Dindo grades in Groups 1 and 2.
Table 5. Incidence of postoperative complications and Clavien-Dindo grades in Groups 1 and 2.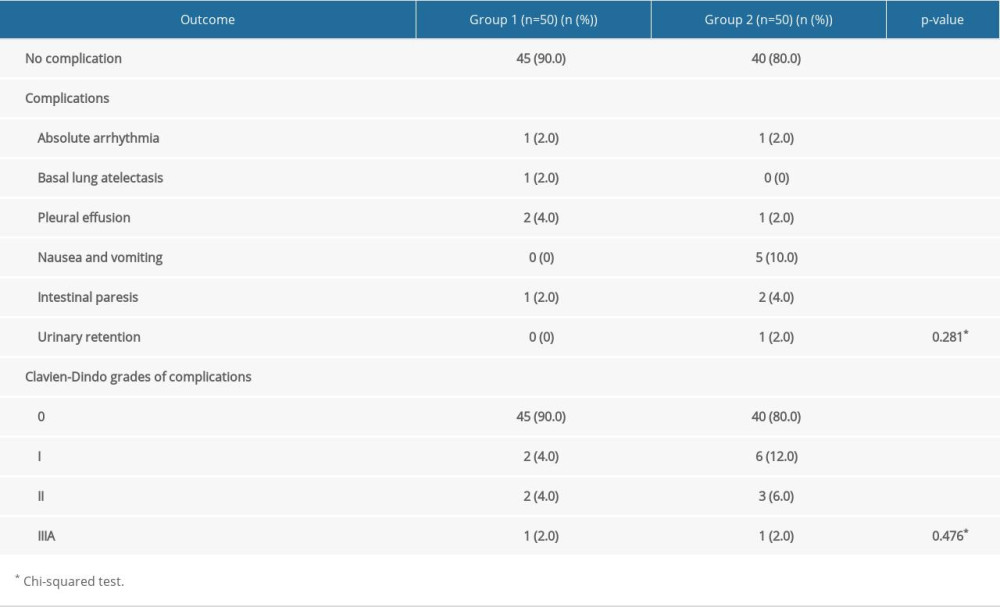
References
1. Ke JJ, Zhan J, Feng XB, A comparison of the effect of total intravenous anaesthesia with propofol and remifentanil and inhalational anaesthesia with isoflurane on the release of pro- and anti-inflammatory cytokines in patients undergoing open cholecystectomy: Anaesth Intensive Care, 2008; 36(1); 74-78
2. Kirdak T, Uysal E, Sezgin E, Inflammatory response markers in rats undergoing abdominal surgical procedures: Ann Gastroenterol, 2020; 33; 528-35
3. Bown MJ, Nicholson ML, Bell PRF, Sayers RD, The systemic inflammatory response syndrome, organ failure, and mortality after abdominal aortic aneurysm repair: J Vasc Surg, 2003; 37; 600-6
4. Kelly KJ, Greenblatt DY, Wan Y, Risk stratification for distal pancreatectomy utilizing ACS-NSQIP: Preoperative factors predict morbidity and mortality: J Gastrointest Surg, 2011; 15(2); 250-59
5. Alazawi W, Pirmadjid N, Lahiri R, Bhattacharya S, Inflammatory and immune responses to surgery and their clinical impact: Ann Surg, 2016; 264(1); 73-80
6. Meisner M, Tschaikowsky K, Hutzler A, Postoperative plasma concentrations of procalcitonin after different types of surgery: Intensive Care Med, 1998; 24(7); 680-84
7. Cruickshank AM, Fraser WD, Burns HJ, Response of serum interleukin-6 in patients undergoing elective surgery of varying severity: Clin Sci (Lond), 1990; 79; 161-65
8. Watt DG, Horgan PG, McMillan DC, Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: A systematic review: Surgery, 2015; 157(2); 362-80
9. Weibel S, Jokinen J, Pace NL, Efficacy and safety of intravenous lidocaine for postoperative analgesia and recovery after surgery: A systematic review with trial sequential analysis: Br J Anaesth, 2016; 116(6); 770-83
10. Pirie K, Traer E, Finniss D, Myles PS, Riedel B, Current approaches to acute postoperative pain management after major abdominal surgery: A narrative review and future directions: Br J Anaesth, 2022; 129(3); 378-93
11. Bugada D, Lorini LF, Lavand’homme P, Opioid free anesthesia: Evidence for short and long-term outcome: Minerva Anestesiol, 2021; 87(2); 230-37
12. Hung KC, Chu CC, Hsing CH, Association between perioperative intravenous lidocaine and subjective quality of recovery: A meta-analysis of randomized controlled trials: J Clin Anesth, 2021; 75; 110521
13. Lauwick S, Kim DJ, Michelagnoli G, Intraoperative infusion of lidocaine reduces postoperative fentanyl requirements in patients undergoing laparoscopic cholecystectomy: Can J Anaesth, 2008; 55(11); 754-60
14. De Oliveira GS, Duncan K, Fitzgerald P, Systemic lidocaine to improve quality of recovery after laparoscopic bariatric surgery: A randomized double-blinded placebo-controlled trial: Obes Surg, 2014; 24(2); 212-18
15. Marret E, Rolin M, Beaussier M, Bonnet F, Meta-analysis of intravenous lidocaine and postoperative recovery after abdominal surgery: Br J Surg, 2008; 95(11); 1331-38
16. Sun Y, Li T, Wang N, Yun Y, Gang TJ, Perioperative systemic lidocaine for postoperative analgesia and recovery after abdominal surgery: A meta-analysis of randomized controlled trials: Dis Colon Rectum, 2012; 55(11); 1183-94
17. Lee IWS, Schraag S, The Use of Intravenous Lidocaine in Perioperative Medicine: Anaesthetic, Analgesic and Immune-Modulatory Aspects: J Clin Med, 2022; 11(12); 3543
18. Kim MH, Lee KY, Park S, Effects of systemic lidocaine versus magnesium administration on postoperative functional recovery and chronic pain in patients undergoing breast cancer surgery: A prospective, randomized, double-blind, comparative clinical trial: PLoS One, 2017; 12(3); e0173026
19. Herroeder S, Pecher S, Schönherr ME, Systemic lidocaine shortens length of hospital stay after colorectal surgery: Ann Surg, 2007; 246(2); 192-200
20. Yardeni IZ, Beilin B, Mayburd E, The effect of perioperative intravenous lidocaine on postoperative pain and immune function: Anesth Analg, 2009; 109(5); 1464-69
21. Beaussier M, Delbos A, Maurice-Szamburski A, Perioperative use of intravenous lidocaine: Drugs, 2018; 78(12); 1229-46
22. Song X, Sun Y, Zhang X, Effect of perioperative intravenous lidocaine infusion on postoperative recovery following laparoscopic cholecystectomy – a randomized controlled trial: Int J Surg, 2017; 45; 8-13
23. Kuo CP, Jao SW, Chen KM, Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain and bowel function in patients undergoing colonic surgery: Br J Anaesth, 2006; 97(5); 640-46
24. Sridhar P, Sistla SC, Ali SM, Karthikeyan VS, Effect of intravenous lignocaine on perioperative stress response and post-surgical ileus in elective open abdominal surgeries: A double-blind randomized controlled trial: ANZ J Surg, 2015; 85(6); 425-29
25. Kranke P, Jokinen J, Pace NL, Continuous intravenous perioperative lidocaine infusion for postoperative pain and recovery: Cochrane Database Syst Rev, 2015; 7; CD009642
26. Aoki Y, Aoshima Y, Atsumi K, Perioperative amino acid infusion for preventing hypothermia and improving clinical outcomes during surgery under general anesthesia: A systematic review and meta-analysis: Anesth Analg, 2017; 125(3); 793-802
27. Chandrasekaran TV, Morgan RN, Mason RA, Nutrient induced thermogenesis during major colorectal excision – a pilot study: Colorectal Dis, 2005; 7(1); 74-78
28. Selldén E, Brundin T, Wahren J, Augmented thermic effect of amino acids under general anaesthesia: A mechanism useful for prevention of anaesthesia-induced hypothermia: Clin Sci (Lond), 1994; 86(5); 611-18
29. Jabłońska B, Mrowiec S, The role of immunonutrition in patients undergoing pancreaticoduodenectomy: Nutrients, 2020; 12; 2547
30. Shu XL, Yu TT, Kang K, Zhao J, Effects of glutamine on markers of intestinal inflammatory response and mucosal permeability in abdominal surgery patients: A meta-analysis: Exp Ther Med, 2016; 12(6); 3499-506
31. Norouzi M, Nadjarzadeh A, Maleki M, The effects of preoperative supplementation with a combination of beta-hydroxy-beta-methylbutyrate, arginine, and glutamine on inflammatory and hematological markers of patients with heart surgery: A randomized controlled trial: BMC Surgery, 2002; 22(1); 51
32. De Oliveira GS, Castro-Alves LJ, Khan JH, McCarthy RJ, Perioperative systemic magnesium to minimize postoperative pain: A meta-analysis of randomized controlled trials: Anesthesiology, 2013; 119(1); 178-90
33. Mendonça FT, Pellizzaro D, Grossi BJ, Synergistic effect of the association between lidocaine and magnesium sulfate on peri-operative pain after mastectomy: A randomised, double-blind trial: Eur J Anaesthesiol, 2020; 37(3); 224-34
34. Albrecht E, Kirkham KR, Liu SS, Brull R, Peri-operative intravenous administration of magnesium sulphate and postoperative pain: A meta-analysis: Anaesthesia, 2013; 68(1); 79-90
35. Lu CY, Shih YL, Sun LC, The inflammatory modulation effect of glutamine-enriched total parenteral nutrition in postoperative gastrointestinal cancer patients: Am Surg, 2011; 77(1); 59-64
36. Saadawy IM, Kaki AM, Abd El Latif AA, Lidocaine vs. magnesium: Effect on analgesia after a laparoscopic cholecystectomy: Acta Anaesthesiol Scand, 2010; 54(5); 549-56
37. Mojtahedzadeh M, Chelkeba L, Ranjvar-Shahrivar M, Randomized trial of the effect of magnesium sulfate continuous infusion on IL-6 and CRP serum levels following abdominal aortic aneurysm surgery: Iran J Pharm Res, 2016; 15(4); 951-56
38. Aryana P, Rajaei S, Bagheri A, Acute effect of intravenous administration of magnesium sulfate on serum levels of interleukin-6 and tumor necrosis factor-α in patients undergoing elective coronary bypass graft with cardiopulmonary bypass: Anesth Pain Med, 2014; 4(3); e16316
39. Agencija za lekove i medicinska sredstva Srbije, 2013 Available online at: [in Bosnian]https://www.alims.gov.rs/doc_file/lekovi/pil/515-01-01282-18-001.pdf
40. Katayama H, Kurokawa Y, Nakamura K, Extended Clavien-Dindo classification of surgical complications: Japan Clinical Oncology Group postoperative complications criteria: Surg Today, 2016; 46(6); 668-85
41. Oberhofer D, Juras J, Pavicic AM, Comparison of C-reactive protein and procalcitonin as predictors of postoperative infectious complications after elective colorectal surgery: Croat Med J, 2012; 53; 612-19
42. Santonocito C, De Loecker I, Donadello K, C-reactive protein kinetics after major surgery: Anesth Analg, 2014; 119(3); 624-29
43. Meyer ZC, Schreinemakers JM, Mulder PG, The role of C-reactive protein and the SOFA score as parameter for clinical decision making in surgical patients during the Intensive Care Unit course: PLoS One, 2013; 8(2); e55964
44. Gans SL, Atema JJ, van Dieren S, Diagnostic value of C-reactive protein to rule out infectious complications after major abdominal surgery: A systematic review and meta-analysis: Int J Colorectal Dis, 2015; 30(7); 861-73
45. Cousin F, Ortega-Deballon P, Bourredjem A, Diagnostic accuracy of procalcitonin and C-reactive protein for the early diagnosis of intra-abdominal infection after elective colorectal surgery: A meta-analysis: Ann Surg, 2016; 264(2); 252-56
46. Domínguez-Comesaña E, Estevez-Fernández SM, López-Gómez V, Procalcitonin and C-reactive protein as early markers of postoperative intra-abdominal infection in patients operated on colorectal cancer: Int J Colorectal Dis, 2017; 32(12); 1771-74
47. Giaccaglia V, Salvi PF, Antonelli MS, Procalcitonin reveals early dehiscence in colorectal surgery: The PREDICS study: Ann Surg, 2016; 263(5); 967-72
48. Lin MT, Kung SP, Yeh SL, Glutamine-supplemented total parenteral nutrition attenuates plasma interleukin-6 in surgical patients with lower disease severity: World J Gastroenterol, 2005; 11(39); 6197-201
49. Lu CY, Shih YL, Sun LC, The inflammatory modulation effect of glutamine-enriched total parenteral nutrition in postoperative gastrointestinal cancer patients: Am Surg, 2011; 77(1); 59-64
50. Richard V, Dahiya D, Kaman L, Raj P, Behera A, Effect of perioperative glutamine administration on C-reactive protein and liver function tests in patients undergoing hepatic resection: Pol Przegl Chir, 2014; 86(1); 11-16
51. Yeh CN, Lee HL, Chiang KC, The role of parenteral glutamine supplement for surgical patient perioperatively: Result of a single center, prospective and controlled study: Langenbecks Arch Surg, 2008; 393(6); 849-55
52. Choi GJ, Kang H, Ahn EJ, Clinical efficacy of intravenous lidocaine for thyroidectomy: A prospective, randomized, double-blind, placebo-controlled trial: World J Surg, 2016; 40(12); 2941-47
53. Sessler DI, Central thermoregulatory inhibition by general anesthesia: Anesthesiology, 1991; 75(4); 557-59
54. Sessler DI, Mild perioperative hypothermia: N Engl J Med, 1997; 336; 1730-37
55. Vigneault L, Turgeon AF, Côté D, Perioperative intravenous lidocaine infusion for postoperative pain control: A meta-analysis of randomized controlled trials: Can J Anaesth, 2011; 58(1); 22-37
56. Nicholson G, Hall GM, Effects of anaesthesia on the inflammatory response to injury: Curr Opin Anaesthesiol, 2011; 24(4); 370-74
57. McCarthy GC, Megalla SA, Habib AS, Impact of intravenous lidocaine infusion on postoperative analgesia and recovery from surgery: A systematic review of randomized controlled trials: Drugs, 2010; 70(9); 1149-63
58. Weibel S, Jelting Y, Pace NL, Continuous intravenous perioperative lidocaine infusion for postoperative pain and recovery in adults: Cochrane Database Syst Rev, 2018; 6(6); CD009642
59. Koppert W, Weigand M, Neumann F, Perioperative intravenous lidocaine has preventive effects on postoperative pain and morphine consumption after major abdominal surgery: Anesth Analg, 2004; 98(4); 1050-55
Figures
 Figure 1. Postoperative WBC, neutrophil, lymphocyte, and PLT counts (109/l) (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test).
Figure 1. Postoperative WBC, neutrophil, lymphocyte, and PLT counts (109/l) (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test). Figure 2. Postoperative levels of fibrinogen (g/l), CRP (mg/l), PCT (ng/ml) and IL-6 (pg/ml) (statistically significant difference is marked with an asterisk (P<0.001); Mann-Whitney U test).
Figure 2. Postoperative levels of fibrinogen (g/l), CRP (mg/l), PCT (ng/ml) and IL-6 (pg/ml) (statistically significant difference is marked with an asterisk (P<0.001); Mann-Whitney U test). Figure 3. Postoperative axillary BT (°C) and NRS (numeric rating scale) scores (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test).
Figure 3. Postoperative axillary BT (°C) and NRS (numeric rating scale) scores (statistically significant differences are indicated by an asterisk (P<0.001); Mann-Whitney U test). Tables
 Table 1. Preoperative parameters in Group 1 and Group 2 (statistically significant differences are in bold; P<0.05).
Table 1. Preoperative parameters in Group 1 and Group 2 (statistically significant differences are in bold; P<0.05). Table 2. Comorbidities in Group 1 and Group 2.
Table 2. Comorbidities in Group 1 and Group 2. Table 3. Intraoperative parameters of Group 1 and 2 (statistically significant differences are in bold; P<0.05).
Table 3. Intraoperative parameters of Group 1 and 2 (statistically significant differences are in bold; P<0.05). Table 4. ROC curve analysis and AUC calculation of the postoperative statistically significant parameters (excluding the PLT count) (statistically significant differences are in bold; P<0.05).
Table 4. ROC curve analysis and AUC calculation of the postoperative statistically significant parameters (excluding the PLT count) (statistically significant differences are in bold; P<0.05). Table 5. Incidence of postoperative complications and Clavien-Dindo grades in Groups 1 and 2.
Table 5. Incidence of postoperative complications and Clavien-Dindo grades in Groups 1 and 2. Table 1. Preoperative parameters in Group 1 and Group 2 (statistically significant differences are in bold; P<0.05).
Table 1. Preoperative parameters in Group 1 and Group 2 (statistically significant differences are in bold; P<0.05). Table 2. Comorbidities in Group 1 and Group 2.
Table 2. Comorbidities in Group 1 and Group 2. Table 3. Intraoperative parameters of Group 1 and 2 (statistically significant differences are in bold; P<0.05).
Table 3. Intraoperative parameters of Group 1 and 2 (statistically significant differences are in bold; P<0.05). Table 4. ROC curve analysis and AUC calculation of the postoperative statistically significant parameters (excluding the PLT count) (statistically significant differences are in bold; P<0.05).
Table 4. ROC curve analysis and AUC calculation of the postoperative statistically significant parameters (excluding the PLT count) (statistically significant differences are in bold; P<0.05). Table 5. Incidence of postoperative complications and Clavien-Dindo grades in Groups 1 and 2.
Table 5. Incidence of postoperative complications and Clavien-Dindo grades in Groups 1 and 2. In Press
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








