26 May 2023: Lab/In Vitro Research
Preparation, Physicochemical Properties, and Structural Characterization of Resistant Starch-Aspirin-Loaded Microspheres
Qilin Zhou12ACDE, Dan Feng3DFG, Haiyan Yang3CD, Fei Luo3B, Yi Zhou2C, Zizhen Yin2B, Yongjian SunDOI: 10.12659/MSM.939711
Med Sci Monit 2023; 29:e939711
Abstract
BACKGROUND: Resistant starch is a novel type of dietary fiber that can be considered as a natural polymer carrier with potential development prospects in the field of oral colonic release preparations since it can be degraded by bacteria in the large intestine.
MATERIAL AND METHODS: In this study, oral resistant starch-drug-loaded microspheres were prepared by spraydrying, and the response surface method was used to optimize the process based on the encapsulation efficiency.
RESULTS: The optimal preparation process conditions for the resistant starch-aspirin-loaded microspheres were as follows: core material: wall material ratio of 1: 1.98, chitosan solution concentration of 1.98%, and spray drying air inlet temperature of 130.45°C resulted in a reliable entrapment efficiency of 68.96%. Infrared spectroscopy analysis indicated that the encapsulated aspirin-starch microspheres did not differ significantly from the original resistant starch material. The ultrastructure of the drug-loaded microspheres was evenly wrapped with the capsule core and appeared as smooth spheres. The combination of resistant starch, aspirin, and chitosan resulted in a cross-linking reaction that reduced the overall gelatinization temperature compared with the original starch material alone. The light transmittance of the drug-loaded microspheres was slightly higher than that of the original resistant starch, while digestibility was similar to that of the resistant starch, indicating that the release would exist in the environment of the large intestine.
CONCLUSIONS: This study provides pivotal insights into the development of resistant starch in the field of colonic release preparations.
Keywords: Drug Carriers, Microspheres, resistant starch, spray drying, Chitosan, Starch, Temperature, Particle Size
Background
Resistant starch (also known as enzyme-resistant starch) cannot be digested or absorbed in the small intestine, but these processes can occur via bacteria in the large intestine [1]. The many health benefits of these starches include aiding digestion [2], reducing serum lipids [2], decreasing inflammation [3], increasing insulin sensitivity, and decreasing the risk of colon cancer [4,5]. These positive physiological benefits have resulted in the widespread use of resistant starch as an additive in foods and healthcare products [6,7], although its medicinal use has not been extensively studied. The recalcitrant nature of these starches also suggests that they could function as microcapsule carriers. The combined effects of slow release in the stomach and small intestine and rapid turnover in the large intestine can be used to exert a systemic therapeutic effect and has broad development prospects in other drug-related fields, such as targeted preparations.
The current methods used for the preparation of microspheres and microcapsules include spray drying, emulsification, extrusion, and complex coacervation. The emulsification method is widely used, and raw material mixtures are easy to control [8]. The extrusion method is also widely used, and its advantages include simple operation and mild reaction conditions, suitable for heat-sensitive preparations. In contrast, spray drying is less used because of technical difficulties of large-scale production. However, this method has yielded excellent results when applied to food and health products and most likely can be used for the encapsulation of pharmaceutical preparations [9,10].
Resistant starch occurs in 1 or more of 4 forms [11]. In the present study, resistant corn starch II was used as the raw material and chitosan as the biological adhesive to explore drug loading via the spraydrying method. Aspirin, a drug that hasoften been loaded as enteric-coated dosage forms [12], was encapsulated in the present study. We evaluated the drug encapsulation efficiency of this resistant starch and optimized the process using response surface analysis. Our results indicated that this natural polymer-based microcapsule drug carrier was superior to synthetic polymer and traditional drug carriers for colonic release formulations and provides a theoretical basis for the use of the combination of spray drying and resistant starch carriers for drug development.
Material and Methods
MATERIALS:
Resistant (corn) starch (Xian Quanao Biotech, Xian, China), aspirin (Gesheng, Jinan, China), and chitosan (Aokang, Xian, China) were all of analytical grade, and α-amylase was obtained from Ryon Tech (Shanghai, China).
MICROSPHERE PREPARATION:
A 1% aqueous solution of corn starch was combined with chitosan and aspirin and stirred at room temperature until all materials were completely dissolved. The material was spray dried at 60°C using a 7.0-μm nozzle and a range of loading rates (4–8 mL/min) and inlet air temperatures (120–140°C). The material was passed through an 80-mesh sieve, allowed to dry completely, and then stored desiccated and protected from light for further experiments. The process was evaluated as previously described, with some modifications [13,14]. Briefly, the aspirin core material: starch wall material ratio (core: wall ratio), mass concentration of chitosan aqueous solution (chitosan concentration), drying inlet temperature, and spray drying injection rate were all varied as gradient inspection indicators to optimize the process parameters (Table 1).
ENCAPSULATION EFFICIENCY DETERMINATIONS:
The encapsulation ratiowas determined by adding the aspirin-loaded microspheres to 0.1 M HCl and ultrasonicating (Kunshan Ultrasonic Instruments, Kunshan, China) for 15 min at 300 W and 40 kHz. The solution was then filtered through a 0.45-μm microporous membrane. The aspirin content was determined using high performance liquid chromatography (Agilent, Santa Clara, CA, USA) with a Zorbax SB-Aq column (250×4.6 mm, 5 μm) using an isocratic mobile phase of methanol: waterratio of 70: 30 at 1 mL/minflow rate with a column temperature of 30 °C. The injection volume was 10 μL, and the eluate was monitored at 220 nm. The encapsulation ratio (ER) was calculated as:
where m1 represents the measured aspirin content (mg) and M represents the theoretical aspirin dosage (mg).
RESPONSE SURFACE METHOD OPTIMIZATION DESIGN AND VALIDATION (BOX-BEHNKEN):
The single-factor test results indicated that 3 factors greatly influenced the encapsulation efficiency. We used these factors for response surface testing using a Box-Behnken design (Table 2). The injection speed was selected as 6 mL/min, and all experiments were repeated 3 times. Design Expert software 8.0.6.1 (Stat-Ease, Minneapolis, MN, USA) was used to perform variance and quadratic polynomial regression fitting of the response values.
STRUCTURAL CHARACTERIZATION:
As previously reported [15–17], we performed the structural analysis of drug-loaded microspheres in the optimal conditions based on encapsulation ratio.
FOURIER TRANSFORM INFRARED SPECTROSCOPY:
For fourier transform infrared spectroscopy (FTIR), dried microcapsule preparations were ground into powders and pressed into KBr tablets and scanned from 400 to 4000 cm−1 using a fourier transform infrared spectrometer (Nicolet, Wisconsin, USA).
SCANNING ELECTRON MICROSCOPY:
During scanning electron microscopy, the fully dried starch drug-loaded microspheres were fixed on a sample stage and scanned at 20 kV using an acceleration current of 9 mA with a field emission scanning electron microscope (Phenom, the Netherlands).
THERMODYNAMIC ANALYSIS:
Thermodynamic analyses were conducted using differential scanning calorimetry (DSC) using a simultaneous thermal analyzer (NETZSCH,Germany). Aspirin-loaded microspheres were sealed into alumina crucibles and scanned at a heating rate of 10°C/min from 30°C to 220°C to construct endothermic curves for each sample that included the initial temperature (T0) of the drug-loaded microsphere sample peak, peak temperature (TP), peak end temperature (Tc), and phase transition enthalpy (ΔH).
TRANSPARENCY:
Starch-aspirin-loaded microspheres (1%) in distilled water were heated in a boiling water bath with constant stirring until gelatinization and then cooled to room temperature before use. The transparency of the paste was measured by absorbance of the paste at room temperature for 0, 1, 2, 3, and 4 h at a wavelength of 620 nm.
IN VITRO DIGESTION PERFORMANCE ASSAY: An in vitro simulated gastrointestinal digestive system was established as previously described with some modifications [18], and the performances were conducted in according with the official standards from the Chinese Pharmacopoeia 2020. Briefly, 250 mg drug-loaded microspheres were mixed with 15 mL of phosphate buffer pH 5.8 in a 100-mL centrifuge tube. The mixture was centrifuged at 37°C and 160 rpm for 5 min and mixed with 10 mL of α-amylase solution (40000u/L). Then, the digestion was performed for 1, 2, 3, 4, 5 and 6 h. At the end of the digestion periods, samples were diluted with phosphate buffer pH 5.8, and glucose content was determined using DNS colorimetry. Each sample was measured 3 times, and the volume of the original solution was replenished with phosphate buffer after sampling. The amount of digested product (B) and the average digestion rate (v) in the entire system were calculated according to the following equations:
where B is the glucose content produced in the system (mg); m1 is the glucose content in each sample (mg); C is the dilution ratio of the digestive solution; V1 is the volume of the entire system solution (mL); V2 is volume of the digestive solution taken each time (mL); 0.001 is the conversion factor; v is the average digestion rate mg/(g·h−1); m is the sample mass (g); and H is the reaction time (h).
For detecting the release of aspirin, artificial digestive juices (simulated gastric fluid, artificial intestinal juice, and artificial colonic fluid) were purchased from Zikerbio (official standards from the Chinese Pharmacopoeia 2020;Shenzhen, China). In vitro release of aspirin was conducted with the dialysis bag diffusion technique with some modifications [19]. Briefly, 500 mL of artificial digestive juice (simulated gastric fluid, artificial intestinal juice, and artificial colonic fluid) was added into respective round-bottomed flasks as release solution. A dialysis bag with 250 mg drug-loaded microspheres was soaked in the artificial digestive juice, and non-loaded microspheres served as controls. Each round-bottomed flask was placed in the dissolution tester (37°C, 100 r/min). Samples of 5 mL were acquired from the flasks at 30-min intervals, then 5 mL of relevant artificial digestive juice was added to make up the volume. The samples were filtered with a 0.22-micron filter and referenced with control microspheres to measure the absorbance values in a spectrometer (Thermo Fisher Scientific, USA). Based on the absorbance values of different concentrations of standard aspirin in the 3 artificial digestive juices, respectively, theabsorbance values of aspirin release in vitro were converted to concentrations [19]. Then the cumulative release (CR) of aspirin (%) was calculated using the following formula:
whereCi is the cumulative concentration of aspirin in release solution (mg/mL); Vnis the volume of the release solution (mL); and mqis the aspirin content in microspheres (mg).
STATISTICAL ANALYSIS:
All data were processed using GraphPad Prism v8.02. Data are expressed as means±standard errors of the mean. For data that were normally distributed and had homogenous variances, comparisons between 2 groups were carried out using the
Results
SINGLE-FACTOR EXPERIMENTS TO OPTIMIZE ENCAPSULATION EFFICIENCY OF THE ASPIRIN-LOADED MICROSPHERES:
The procedure for the preparation of aspirin-loaded microspheres by spray drying was optimized using 4 single-factor experiments that included varying the core: wallmaterial ratio, chitosan solution concentration, spray drying air inlet temperature, and spray drying injection speed. As the core: wallmaterial ratio of the microspheres was increased, the encapsulation ratio displayed an initial increase and then it decreased. The maximum value (69.93%) was reached at a core: wallmaterial ratio of 1: 2 (Figure 1A). Increasing the chitosan concentration also resulted in an initial increase, followed by a decreased encapsulation efficiency, with a maximal level (70.72%) reached at a chitosan concentration of 2% (Figure 1B). Similarly, as the in-take temperature was increased, the encapsulation efficiency reached a maximal level (70.75%) at 130 °C (Figure 1C). The injection speed also showed a trend of an initial increase followed by a decrease, and the maximum value (70.17%) was reached when the injection speed was 6 mL/min (Figure 1D).
BOX-BEHNKENRESPONSE SURFACE TESTS, MODEL FITTING,AND DESIGN OPTIMIZATION:
The statistical requirements of the Box-Behnken design coupled with the requirements of experimental factors and levels resulted in 17 designed experiments (Table 3). A nonlinear regression equation for encapsulation efficiency was obtained: Y=−2.94A2-2.11B2-1.61C2+0.42AB+0.88AC+0.15BC-0.00375A-0.086B+0.15C+70.21. Based on the equation, we established a model-to-fit parameter for the prediction of the encapsulation ratio. The model had a Pvalue of 0.0001, the lack of fit had a P value of 0.5394, and the coefficient of determination for the model was R2=0.9335 (Table 4), suggesting good model fitting. A quadratic polynomial fitting equation was used to draw contour maps on the 2-dimensional planes and 3-dimensional surface maps of the test factors A, B, and C. The shape of the contour lines reflected interaction strengths, and significant and insignificant interactions were indicated by ovals and circles, respectively. The steepness of the response surface (response surface plots) was directly related to the strength of the influence of a factor on the response value.
The contour maps of the inlet air temperature were all elliptical, suggesting that air temperature interactions with the other factors were significant (Figure 2A–2C). This indicated that the core: wall material ratio as well as the chitosan concentration had great influences on the yields of the drug-loaded microspheres. In particular, as the core: wall material ratio was increased, the response value first increased and then decreased, and the maximal yields were obtained with a ratio of 1: 1.5 to 1: 2.5. An increase in chitosan concentration resulted in an initial increase, followed by a decrease of the encapsulation efficiency. The maximal yields were obtained at chitosan levels of 1.5% to 2.5% (Figure 2A). Similarly, the core: wall material ratios interacted with the spray drying inlet air temperature and temperature ranges of 125°Cto 135°C resulted in maximal encapsulation. This air temperature range resulted in a significant change in encapsulation efficiency for core: wall material ratios of 1: 2.5 to 1: 3. At the core: wall ratio of 1: 1.5 to 1: 2.5 and the inlet air temperature 125°Cto 135°C, the highest point of the response surface was achieved (Figure 2B).
Interactions between chitosan levels and the inlet air temperature demonstrated a direct correlation between temperature and response value that increased and then decreased. Chitosan levels of 1.5% to 2.5% coupled with inlet air temperatures ranging from 125°Cto 135°C resulted in the highest point for the response surface (Figure 2C).
Based on Design Expert software (8.0.6.1), optimization of the experimental conditions were as follows: core: wall material ratio of 1: 1.98, chitosan solution concentration 1.98%, and spray drying air inlet temperature of 130.45 °C,resulting in atheoretical entrapment efficiency of 70.22% (Figure 2D and Table 5). In practice, the entrapment efficiency was 68.96%, and a low error (within 3%) proved the reliability of optimization (Figure 2E).
STRUCTURAL FTIR, SCANNING ELECTRON MICROSCOPY, AND THERMODYNAMIC ANALYSIS OF RESISTANT STARCH-DRUG-LOADED MICROSPHERES:
We then conducted a comprehensive structural analysis for the prepared aspirin-loaded microspheres. We found a stretching vibration peak for associative −OH at 3385 cm−1, a C–H asymmetric stretching vibration absorption peak at 2929 cm−1, and a stretching vibration peak at 1643 cm−1. The 2 −OH absorption peaks for absorbed water indicated an asymmetric stretching vibration absorption peak of C-O-C at 1155 cm−1, a symmetrical vibration absorption peak for C-(OH) at 1020 cm−1, and a symmetric vibration absorption peak at 929 cm−1. There were sugar ring stretching vibration absorption peaks at 860, 763, and 576 cm−1. However, the IR spectra of the prepared drug-loaded microspheres did not change significantly, compared with the proto-resistant starch (Figure 3A), indicating that there was no chemical reaction between the resistant starch and the loaded drug, but only physical cross-linking or adsorption, which can be more conducive to release drug and do not affected drug embedding. An ultrastructural analysis using scanning electron microscopy indicated that the drug-loaded microspheres had smooth particle surfaces and were mostly granules of regular spheres or ellipsoids (Figure 3B). DSC analysis of our microcapsule samples (Table 6) indicated that gelatinization of the drug-loaded microspheres was 55.07°Cto 107.86°C, with an associated enthalpy of 37.40 J/g. In contrast, the gelatinization temperature and enthalpy for the starch material were 38.50°C to 133.39°C and 316.81 J/g, respectively (Table 6). The as-prepared drug-loaded microspheres generated peaks at 73.84°C, while that of proto-resistant starch was 88.33°C (Figure 3C).
PHYSICOCHEMICAL PROPERTIES AND IN VITRO DIGESTION PERFORMANCE OF RESISTANT STARCH DRUG-LOADED MICROSPHERES:
The transparency (transmittance) of the prepared pastes reflects the mutual solubility of the drug-loaded microspheres and water, and this can directly affect its practical use. The transparency of the aspirin-loaded microsphere paste did not significantly differ from that of the original resistant starch material (Figure 4A). The determination curves for transparency of these pastes were strongly correlated (Pearson R=0.98), suggesting that the mutual solubility and swelling were similar.
The in vitro digestion performance of resistant starch-aspirin-loaded microspheres indicated that the quantity of digestive product increased with digestion time (Figure 4B). The average digestion rate decreased significantly in the first 4 h, followed by a slow decline. In addition, increases in the amount of product resulted in greater yields at the same digestion times. The aspirin-loaded material and the source resistant starch material displayed similar digestion curves, which were highly correlated (Figure 4C). The in vitro release test showed that the cumulative release percentage of aspirin in artificial colonic fluid was significantly higher than that in simulated gastric fluid and artificial intestinal juice (Figure 4D). These results indicated that the in vitro digestion performance of starch-drug-loaded microspheres was similar to that of the resistant starch, and the aspirin-loaded microspheres could release aspirin mostly in the environment of the large intestine.
Discussion
Resistant starch has been widely used in foods and health care products owing to its excellent health benefits, although it has not been used in the pharmaceutical field. Interestingly, there are studies of the use of resistant starch as a drug carrier in the treatment of intestinal disease [20–22]. The most common procedures for producing starch microcapsules are emulsification and extrusion; spray drying has not been widely applied, although this method can achieve high homogeneity and encapsulation levels [23,24]. Aspirin is a nonsteroidal anti-inflammatory drug. Because the long-term use of aspirin can result in gastric mucosal injury, it has often been loaded in enteric-coated dosage forms [12]. Chitosan, a biodegradable material that is biocompatible and non-toxic, is the polymer of choice for delivery of the active moieties to the colon owing to its cationic nature that enables strong mucosal attachment [25,26]. Considering that resistant starch is digested or absorbed only in the large intestine, in this study, aspirin-chitosan-resistant starch microspheres were prepared by spray drying, and we utilized single-factor experiments that were optimized using the Box-Behnken design to obtain the most suitable process parameters for the production of aspirin-starch. We confirmed the successful use of spraydrying to construct drug-release microcarriers based on resistant starch. Our single-factor experiments indicated that the encapsulation efficiency displayed initial increases followed by decreases for all our process parameters. Interestingly, starch aqueous solutions resulted in nozzle clogging at sizes <7.0 μm, and encapsulation efficiency was optimal at the larger pore size of 7.0 μm.
We also chose the response surface methodology–Box-Behnkendesign as the optimization model since it possessed a rotatable design and required fewer runs than the full factorial or central composite design [27]. Box and Behnken (1960) developed 3level designs that allowed estimation of a general quadratic model. These designs consisted of 22 factorials in each pair of factors, with all other factors held constant at their mid-level, plus the addition of a few center points. Box-Behnken designs require factors to be varied over 3 levels and they usually require fewer total runs than the central composite design and makes experimentation more economical [28]. The most suitable multivariate regression equation we obtained fit the functional relationships between factors and effect size. As factor levels were increased, the 3-dimensional response surface graphs were altered and displayed elliptical shapes. The initial increases followed by decreases in the response values indicated that the 3 selected factors were effective for maximizing encapsulation.
At the same time, the structure of the obtained resistant starch-drug-loaded microspheres and FTIR measurements for the aspirin-loaded microspheres did not significantly differ from that of the source starch material. These was no characteristic absorption peak for aspirin evident in the spectra, indicating that the resistant starch wall material was combined with the aspirin core and the aspirin molecules were dispersed in the 3-dimensional structure formed by the microspheres. In this way, drug loading can be achieved through physical connections or simple valence bonds in which there is no chemical structure change. The encapsulation of the aspirin also resulted in the conversion of the rough shapes of the original starch material to spheres and ellipsoids, with good encapsulation of the wall material and anevenly wrapped capsule core. The sphericity was relatively compact and stable, and the particle size of the microspheres reached the micron level, which is necessary for microspheres with good release properties [29].
The peak gelatinization temperature for the aspirin-loaded microspheres was slightly lower than that of the original corn starch material, and the gelatinization enthalpy was greatly reduced. The addition of chitosan allowed a cross-linking reaction to occur, which altered the original structure of the resistant starch, thereby reducing the overall gelatinization temperature. FTIR and DSC analysis has shown that there is no interaction between resistant starch and the loaded aspirin [30]. These results indicate that the aspirin-resistant starch combination was successful and can carry aspirin-active drug ingredients [31]. Our initial purpose for the use of aspirin was to explore whether other common drugs can be well combined with the carrier. For example, steroidal anti-inflammatory drugs can be prepared in the manner presented here to exert systemic anti-inflammatory, analgesic, and other therapeutic effects.
The paste preparation of the aspirin-starch microspheres displayed a slightly higher transmittance than the original starch and was most likely related to a lower linear molecule content and elevated aspirin-starch mutual associations. A decreased content of linear molecules weakened the light scattering effect and increased transparency, consistent with a previous study [32]. The number of digested products gradually increased with the extension of digestion time, and the digestion was relatively slow and uniform, indicating controllability and a slow-release character, consistent with an earlier study [33]. It is clear that the use of resistant starch as a carrier material for microcapsules is reliable and can play a leading role in research in the field of colonic release preparations. Resistant starch can also allow the effect of directional and slow release in the stomach and small intestine and fast release in the large intestine. These properties make the resistant starch-drug-loaded microspheres promising in the field of targeted therapy for colorectal diseases.
Conclusions
In summary, the prepared resistant starch-drug-loaded microspheres could delay degradation when passing through the stomach and small intestine. However, it may achieve an effect of fast release of the active ingredients of the preparation in the large intestine. In addition, the drug-loaded microspheres prepared by spray drying also have good encapsulation and drug-loading qualities. This pioneering work can provide a reference for others to continue their research in the field and provide possibilities for clinical applications.
Figures
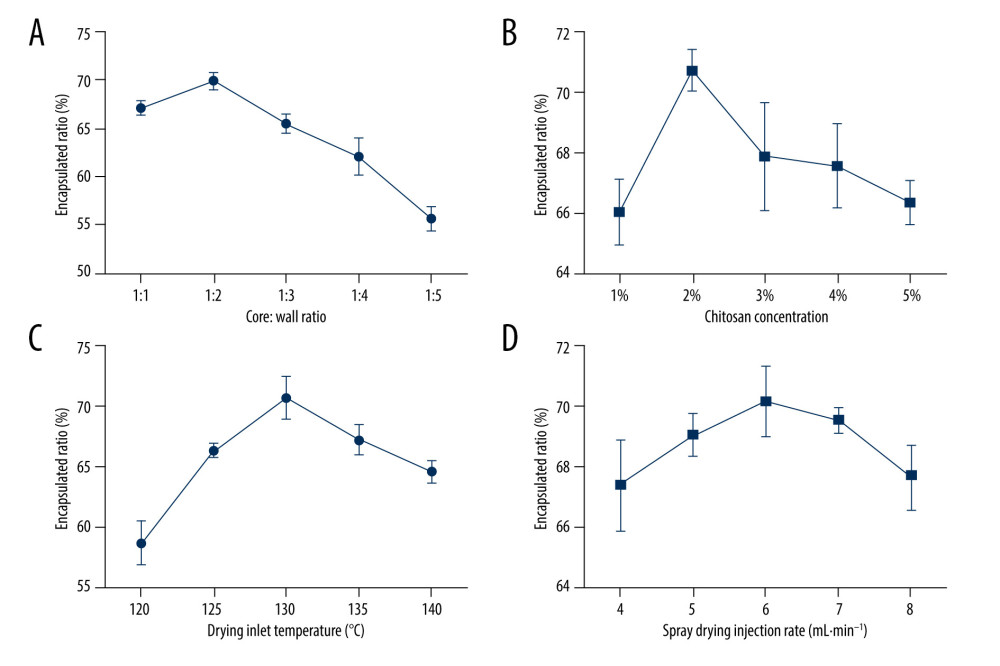 Figure 1. Univariate analysis of the effect of drug-loaded microspheres on the encapsulation efficiency. Influences on the encapsulation efficiency of drug-loaded microspheres by (A) corematerial: wallmaterial ratio, (B) chitosan concentration, (C) drying inlet temperature, and (D) spray drying injection rate.
Figure 1. Univariate analysis of the effect of drug-loaded microspheres on the encapsulation efficiency. Influences on the encapsulation efficiency of drug-loaded microspheres by (A) corematerial: wallmaterial ratio, (B) chitosan concentration, (C) drying inlet temperature, and (D) spray drying injection rate. 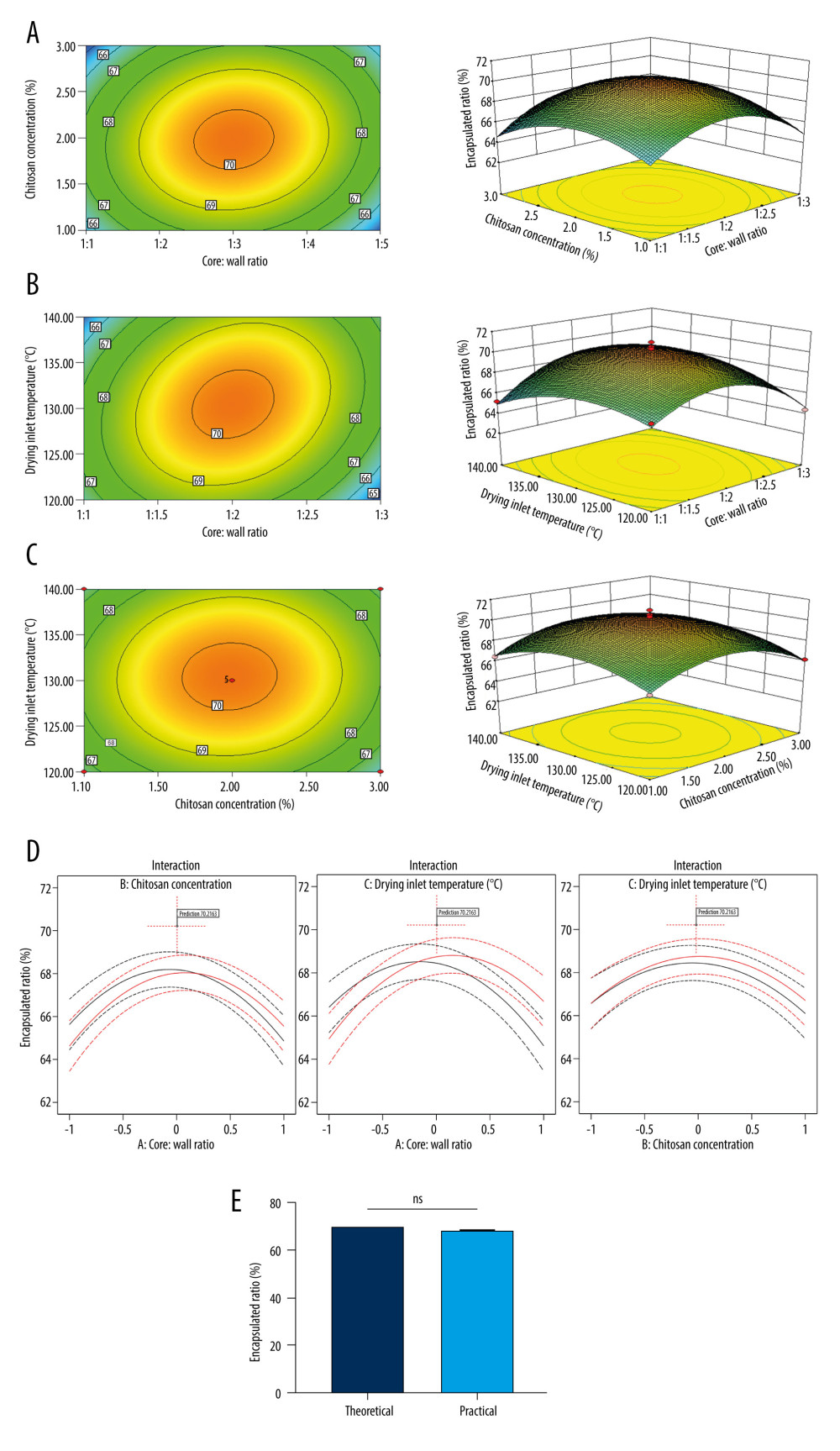 Figure 2. Influencing factors of Box-Behnken response surface test optimization. Contour plots and response surface plots for (A) corematerial: wallmaterial ratios and chitosan concentrations, (B) corematerial: wallmaterial ratio and drying inlet temperature,and (C) chitosan concentration and drying inlet temperature. (D) Optimization of the conditions. (E) Theoretical and practical encapsulated ratios based on the optimized conditions.
Figure 2. Influencing factors of Box-Behnken response surface test optimization. Contour plots and response surface plots for (A) corematerial: wallmaterial ratios and chitosan concentrations, (B) corematerial: wallmaterial ratio and drying inlet temperature,and (C) chitosan concentration and drying inlet temperature. (D) Optimization of the conditions. (E) Theoretical and practical encapsulated ratios based on the optimized conditions. 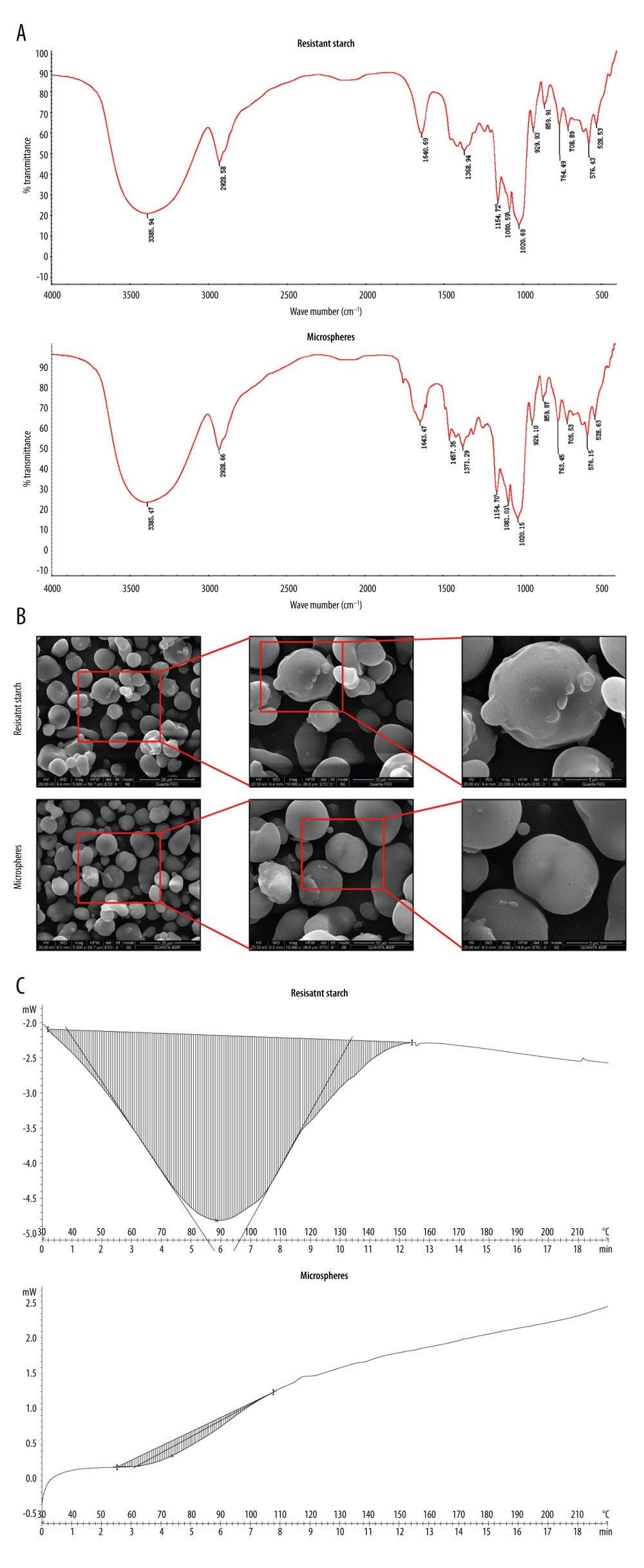 Figure 3. Structural and thermodynamic analysis of drug-loaded microspheres. Analysis of the resistant starch and drug-loaded microspheres. (A) Fourier transform infrared spectroscopy analysis, (B) scanning electron microscopy analysis, and (C) thermodynamic analysis.
Figure 3. Structural and thermodynamic analysis of drug-loaded microspheres. Analysis of the resistant starch and drug-loaded microspheres. (A) Fourier transform infrared spectroscopy analysis, (B) scanning electron microscopy analysis, and (C) thermodynamic analysis. 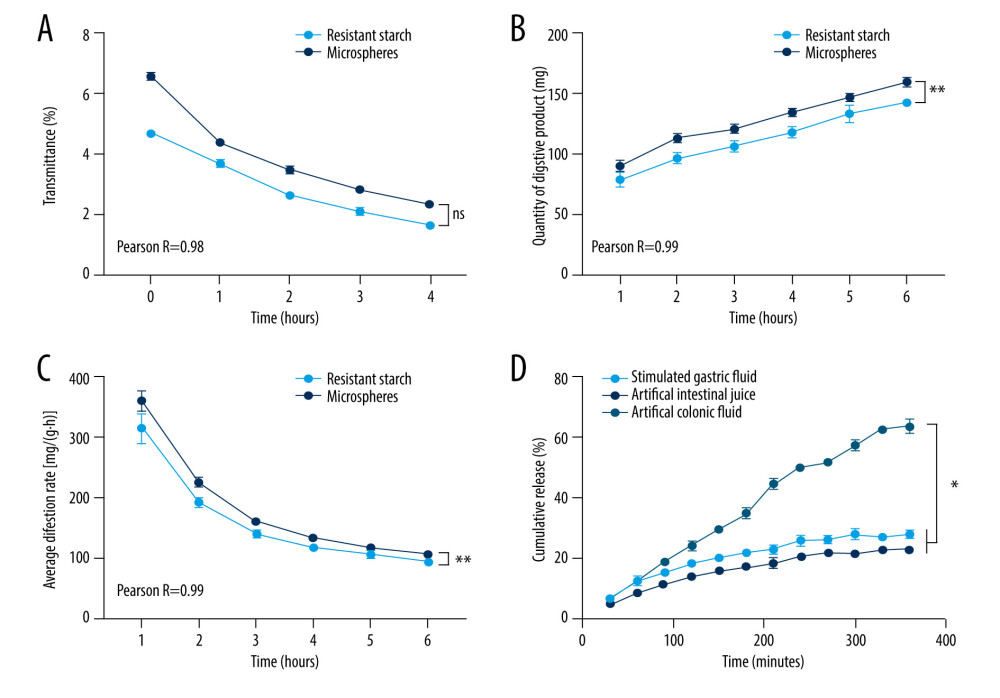 Figure 4. Analysis of physicochemical properties of drug-loaded microspheres. (A) Transmittance of paste preparations, (B) quantity of digested product, and (C) average digestion rate for resistant starch and drug-loaded microspheres. (D) The cumulative release of aspirin in artificial digestive juices (simulated gastric fluid, artificial intestinal juice, and artificial colonic fluid).
Figure 4. Analysis of physicochemical properties of drug-loaded microspheres. (A) Transmittance of paste preparations, (B) quantity of digested product, and (C) average digestion rate for resistant starch and drug-loaded microspheres. (D) The cumulative release of aspirin in artificial digestive juices (simulated gastric fluid, artificial intestinal juice, and artificial colonic fluid). Tables
Table 1. Single-factor experimental design.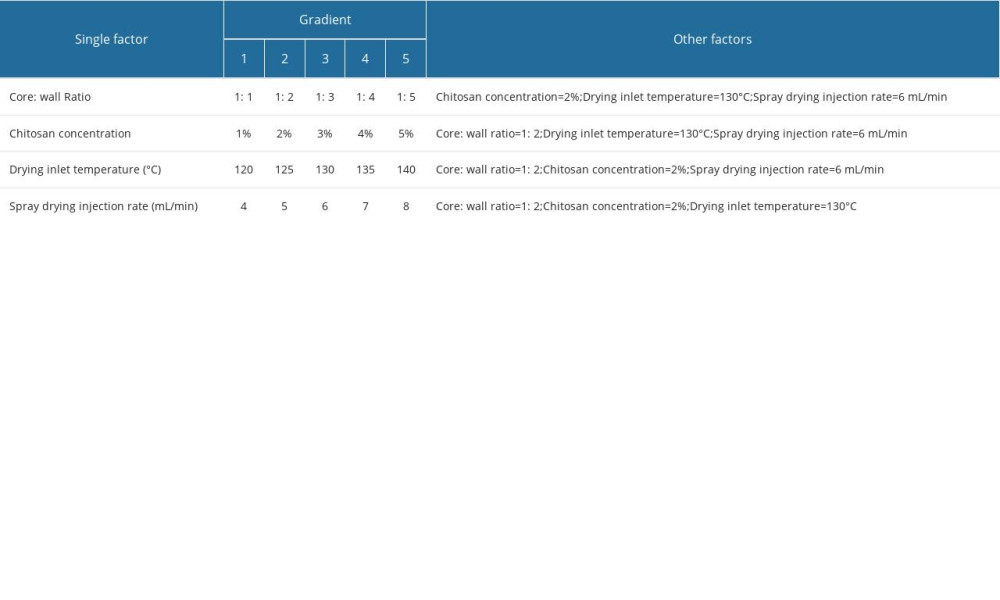 Table 2. Box-Behnken test design factors, levels, and coding.
Table 2. Box-Behnken test design factors, levels, and coding.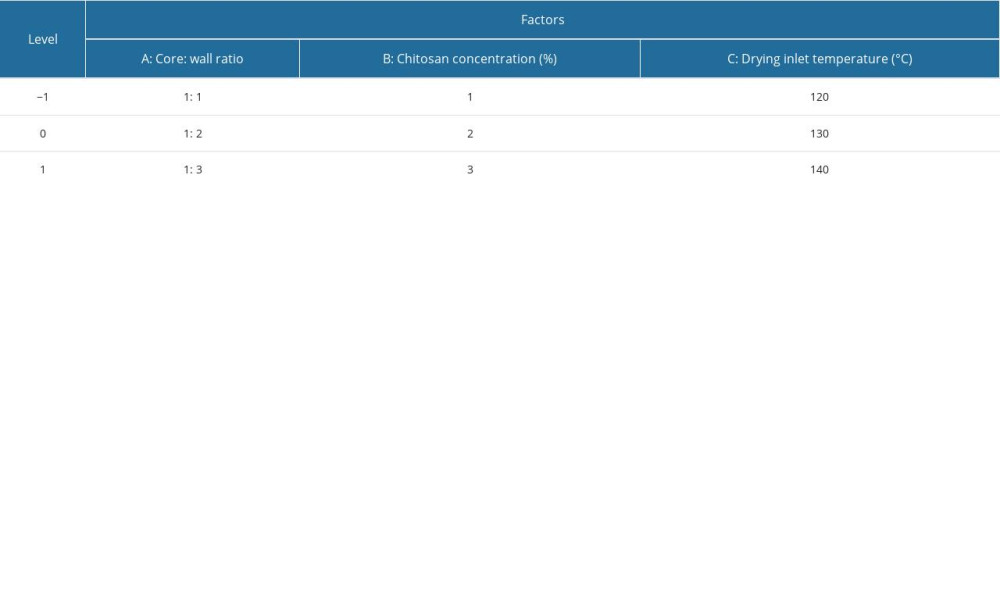 Table 3. Box-Behnken test design and results.
Table 3. Box-Behnken test design and results.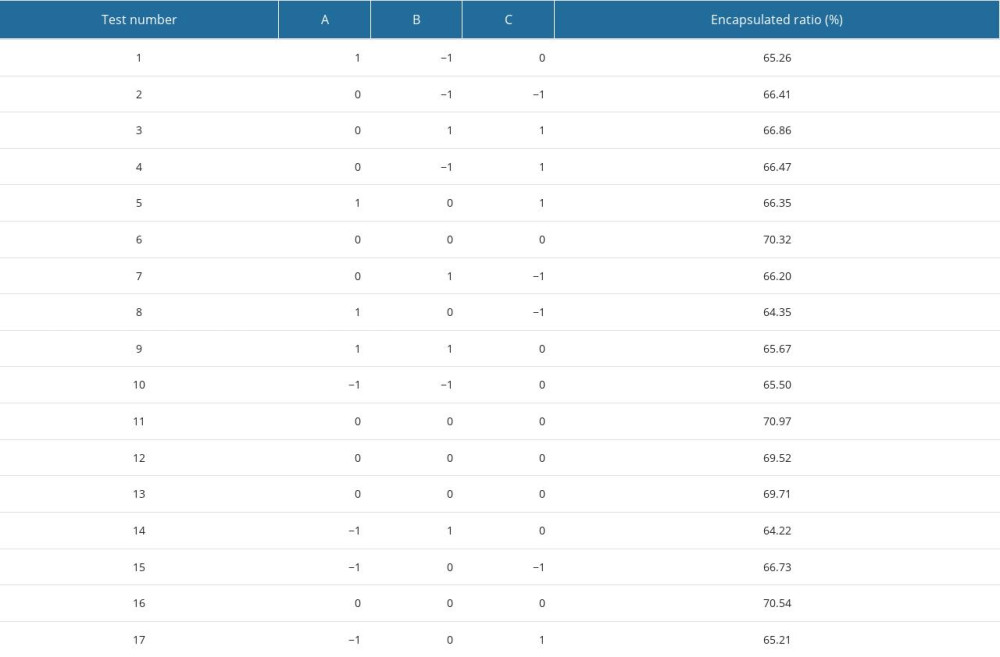 Table 4. Results of analysis of variance.
Table 4. Results of analysis of variance.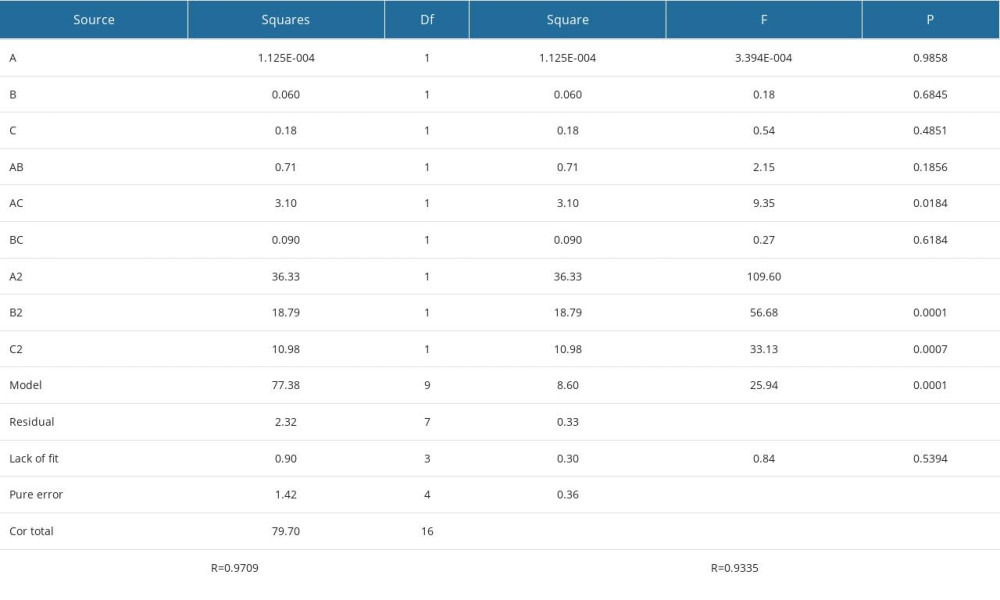 Table 5. Optimization of the conditions, based on Design Expert software.
Table 5. Optimization of the conditions, based on Design Expert software.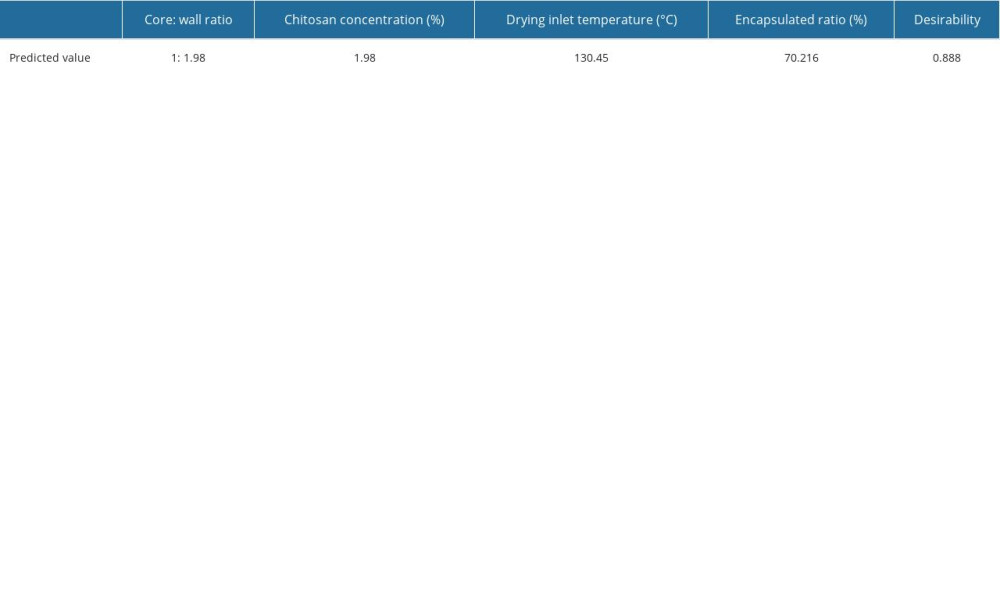 Table 6. Thermodynamic characteristics of resistant starch and drug-loaded microspheres.
Table 6. Thermodynamic characteristics of resistant starch and drug-loaded microspheres.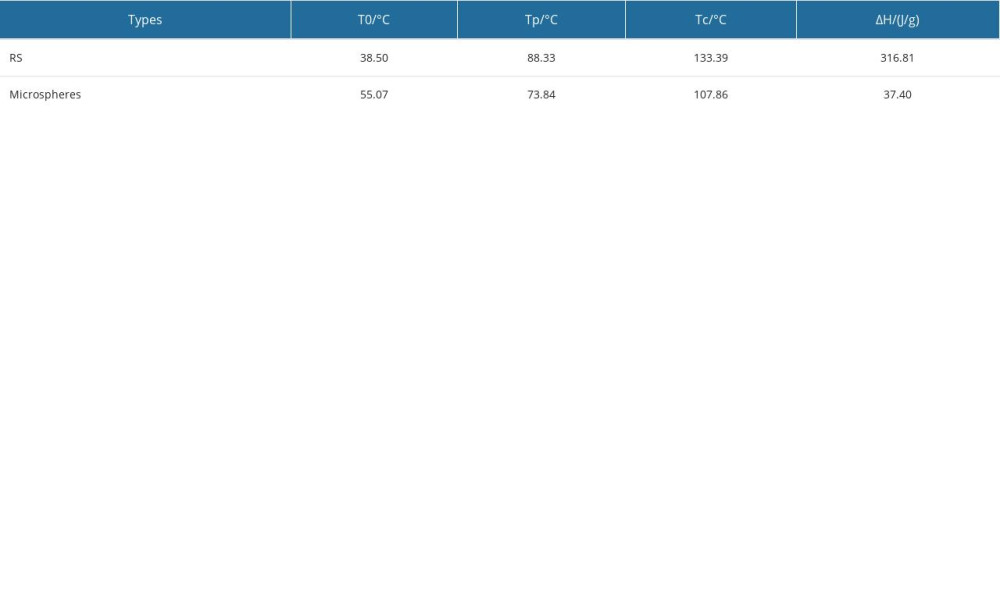
References
1. DeMartino P, Cockburn DW, Resistant starch: impact on the gut microbiome and health: Curr Opin Biotechnol, 2020; 61; 66-71
2. Ma Z, Boye JI, Research advances on structural characterization of resistant starch and its structure-physiological function relationship: A review: Crit Rev Food Sci Nutr, 2018; 58(7); 1059-83
3. Bach Knudsen KE, Laerke HN, Hedemann MS, Impact of diet-modulated butyrate production on intestinal barrier function and inflammation: Nutrients, 2018; 10(10); 1499
4. Hu Y, Li C, Hou Y, Possible regulation of liver glycogen structure through the gut-liver axis by resistant starch: A review: Food Funct, 2021; 12(22); 11154-64
5. Chisenga SM, Workneh TS, Bultosa G, Alimi BA, Progress in research and applications of cassava flour and starch: A review: J Food Sci Technol, 2019; 56(6); 2799-813
6. Lal MK, Tiwari RK, Kumar R: Food Chem, 2021; 359; 129939
7. Hughes RL, Horn WH, Finnegan P, Resistant starch type 2 from wheat reduces postprandial glycemic response with concurrent alterations in gut microbiota composition: Nutrients, 2021; 13(2); 645
8. Comunian TA, Favaro LF, Thomazini M, Echium oil with oxidative stability increased by emulsion preparation in the presence of the phenolic compound sinapic acid followed by dehydration by spray and freeze drying processes: J Food Sci Technol, 2019; 56(3); 1155-64
9. Vaucher A, Dias PCM, Coimbra PT, Microencapsulation of fish oil by casein-pectin complexes and gum arabic microparticles: oxidative stabilisation: J Microencapsul, 2019; 36(5); 459-73
10. Eski A, Demirbag Z, Demir I: J Microencapsul, 2019; 36(1); 1-9
11. Niba LL, Resistant starch: A potential functional food ingredient: Nutrition & Food Science, 2002; 32(2); 62-67
12. Du L, Gao F, Zhang J, Effect of weikang capsule () on aspirin-related gastric and small intestinal mucosal injury: Chin J Integr Med, 2021; 27(8); 621-25
13. Arslan-Tontul S: Probiotics Antimicrob Proteins, 2020; 12(2); 747-55
14. Garcia-Fernandez MJ, Tabary N, Chai F, New multifunctional pharmaceutical excipient in tablet formulation based on citric acid-cyclodextrin polymer: Int J Pharm, 2016; 511(2); 913-20
15. Elfstrand L, Eliasson AC, Wahlgren M, The effect of starch material, encapsulated protein and production conditions on the protein release from starch microspheres: J Pharm Sci, 2009; 98(10); 3802-15
16. Vila A, Sanchez A, Tobio M, Calvo P, Alonso MJ, Design of biodegradable particles for protein delivery: J Control Release, 2002; 78(1–3); 15-24
17. Takeuchi H, Yamamoto H, Kawashima Y, Mucoadhesive nanoparticulate systems for peptide drug delivery: Adv Drug Deliv Rev, 2001; 47(1); 39-54
18. Zhang B, Bai B, Pan Y, Effects of pectin with different molecular weight on gelatinization behavior, textural properties, retrogradation and in vitro digestibility of corn starch: Food Chem, 2018; 264; 58-63
19. Ranjan A, Jha PK, Experiments and modeling of controlled release behavior of commercial and model polymer-drug formulations using dialysis membrane method: Drug Deliv Transl Res, 2020; 10(2); 515-28
20. Schicho A, Pereira PL, Michalik K, Safety and efficacy of transarterial chemoembolization with degradable starch microspheres (DSM-TACE) in the treatment of secondary liver malignancies: Onco Targets Ther, 2018; 11; 345-50
21. Chen F, Cao X, Yu J, Quaternary ammonium groups modified starch microspheres for instant hemorrhage control: Colloids Surf B Biointerfaces, 2017; 159; 937-44
22. Goszczynski TM, Filip-Psurska B, Kempinska K, Hydroxyethyl starch as an effective methotrexate carrier in anticancer therapy: Pharmacol Res Perspect, 2014; 2(3); e00047
23. Shi L, Beamer SK, Yang H, Jaczynski J, Micro-emulsification/encapsulation of krill oil by complex coacervation with krill protein isolated using isoelectric solubilization/precipitation: Food Chem, 2018; 244; 284-91
24. Ghanbarzadeh S, Valizadeh H, Yaqoubi S, Application of spray drying technique for flowability enhancement of divalproex sodium: Drug Res (Stuttg), 2018; 68(3); 168-73
25. Kulkarni N, Jain P, Shindikar A, Advances in the colon-targeted chitosan based multiunit drug delivery systems for the treatment of inflammatory bowel disease: Carbohydr Polym, 2022; 288; 119351
26. Liu Y, Yuan Y, Duan S, Preparation and characterization of chitosan films with three kinds of molecular weight for food packaging: Int J Biol Macromol, 2020; 155; 249-59
27. Nalawade V, Vora A, Box-Behnken design directed optimization for sensitivity assessment of anti-platelet drugs: Drug Dev Ind Pharm, 2019; 45(9); 1515-22
28. Inan S, Mumcu T, Seyhan Bozkurt S, Box-Behnken design for removal of uranium(VI) from aqueous solution using poly(ethylene glycol) based dicationic ionic liquid impregnated chitosan: Turk J Chem, 2020; 44(3); 756-74
29. Yan S, Ren J, Jian Y, Injectable maltodextrin-based micelle/hydrogel composites for simvastatin-controlled release: Biomacromolecules, 2018; 19(12); 4554-64
30. Jaiturong P, Sirithunyalug B, Eitsayeam S, Preparation of glutinous rice starch/polyvinyl alcohol copolymer electrospun fibers for using as a drug delivery carrier: Asian J Pharm Sci, 2018; 13(3); 239-47
31. Li P, He X, Dhital S, Structural and physicochemical properties of granular starches after treatment with debranching enzyme: Carbohydr Polym, 2017; 169; 351-56
32. Chen L, Tian Y, Tong Q, Effect of pullulan on the water distribution, microstructure and textural properties of rice starch gels during cold storage: Food Chem, 2017; 214; 702-9
33. Miyagusuku-Cruzado G, Jimenez-Flores R, Giusti MM, Whey protein addition and its increased light absorption and tinctorial strength of model solutions colored with anthocyanins: J Dairy Sci, 2021; 104(6); 6449-62
Figures
 Figure 1. Univariate analysis of the effect of drug-loaded microspheres on the encapsulation efficiency. Influences on the encapsulation efficiency of drug-loaded microspheres by (A) corematerial: wallmaterial ratio, (B) chitosan concentration, (C) drying inlet temperature, and (D) spray drying injection rate.
Figure 1. Univariate analysis of the effect of drug-loaded microspheres on the encapsulation efficiency. Influences on the encapsulation efficiency of drug-loaded microspheres by (A) corematerial: wallmaterial ratio, (B) chitosan concentration, (C) drying inlet temperature, and (D) spray drying injection rate. Figure 2. Influencing factors of Box-Behnken response surface test optimization. Contour plots and response surface plots for (A) corematerial: wallmaterial ratios and chitosan concentrations, (B) corematerial: wallmaterial ratio and drying inlet temperature,and (C) chitosan concentration and drying inlet temperature. (D) Optimization of the conditions. (E) Theoretical and practical encapsulated ratios based on the optimized conditions.
Figure 2. Influencing factors of Box-Behnken response surface test optimization. Contour plots and response surface plots for (A) corematerial: wallmaterial ratios and chitosan concentrations, (B) corematerial: wallmaterial ratio and drying inlet temperature,and (C) chitosan concentration and drying inlet temperature. (D) Optimization of the conditions. (E) Theoretical and practical encapsulated ratios based on the optimized conditions. Figure 3. Structural and thermodynamic analysis of drug-loaded microspheres. Analysis of the resistant starch and drug-loaded microspheres. (A) Fourier transform infrared spectroscopy analysis, (B) scanning electron microscopy analysis, and (C) thermodynamic analysis.
Figure 3. Structural and thermodynamic analysis of drug-loaded microspheres. Analysis of the resistant starch and drug-loaded microspheres. (A) Fourier transform infrared spectroscopy analysis, (B) scanning electron microscopy analysis, and (C) thermodynamic analysis. Figure 4. Analysis of physicochemical properties of drug-loaded microspheres. (A) Transmittance of paste preparations, (B) quantity of digested product, and (C) average digestion rate for resistant starch and drug-loaded microspheres. (D) The cumulative release of aspirin in artificial digestive juices (simulated gastric fluid, artificial intestinal juice, and artificial colonic fluid).
Figure 4. Analysis of physicochemical properties of drug-loaded microspheres. (A) Transmittance of paste preparations, (B) quantity of digested product, and (C) average digestion rate for resistant starch and drug-loaded microspheres. (D) The cumulative release of aspirin in artificial digestive juices (simulated gastric fluid, artificial intestinal juice, and artificial colonic fluid). Tables
 Table 1. Single-factor experimental design.
Table 1. Single-factor experimental design. Table 2. Box-Behnken test design factors, levels, and coding.
Table 2. Box-Behnken test design factors, levels, and coding. Table 3. Box-Behnken test design and results.
Table 3. Box-Behnken test design and results. Table 4. Results of analysis of variance.
Table 4. Results of analysis of variance. Table 5. Optimization of the conditions, based on Design Expert software.
Table 5. Optimization of the conditions, based on Design Expert software. Table 6. Thermodynamic characteristics of resistant starch and drug-loaded microspheres.
Table 6. Thermodynamic characteristics of resistant starch and drug-loaded microspheres. Table 1. Single-factor experimental design.
Table 1. Single-factor experimental design. Table 2. Box-Behnken test design factors, levels, and coding.
Table 2. Box-Behnken test design factors, levels, and coding. Table 3. Box-Behnken test design and results.
Table 3. Box-Behnken test design and results. Table 4. Results of analysis of variance.
Table 4. Results of analysis of variance. Table 5. Optimization of the conditions, based on Design Expert software.
Table 5. Optimization of the conditions, based on Design Expert software. Table 6. Thermodynamic characteristics of resistant starch and drug-loaded microspheres.
Table 6. Thermodynamic characteristics of resistant starch and drug-loaded microspheres. In Press
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
11 Mar 2024 : Clinical Research
Comparison of Effects of Sugammadex and Neostigmine on Postoperative Neuromuscular Blockade Recovery in Pat...Med Sci Monit In Press; DOI: 10.12659/MSM.942773
12 Mar 2024 : Clinical Research
Comparing Neuromuscular Blockade Measurement Between Upper Arm (TOF Cuff®) and Eyelid (TOF Scan®) Using Miv...Med Sci Monit In Press; DOI: 10.12659/MSM.943630
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








