27 June 2023: Review Articles
A Review of the Mechanism of Action and Clinical Applications of Osmotic Dilators for Cervical Ripening in the Induction of Labor and in Gynecology Procedures
Maciej W. SochaDOI: 10.12659/MSM.940127
Med Sci Monit 2023; 29:e940127
Abstract
ABSTRACT: Cervical ripening is defined as the significant softening of the cervical tissue that usually begins before the onset of contractions during labor, and is required for cervical dilation and delivery. Osmotic dilators are medical implements that dilate the uterine cervix by increasing in size as they absorb fluid from the surrounding tissue. This article aims to review the mechanisms and applications of osmotic dilators for cervical ripening in the induction of labor and in gynecology procedures.
Keywords: Cervical Ripening, Labor, Induced, Laminaria, Obstetrics, Dilapan, Pregnancy, Female, Humans, Gynecology, Abortion, Induced, Dilatation
Background
Despite the fact that the human cervix is a small part of the uterus, it plays an important role in various processes [1]. During the course of pregnancy, the cervix maintains a physical barrier, so the fetal development remains undisturbed. It is also actively involved in parturition by becoming soft and susceptible so the fetus can be delivered [2]. In a non-pregnant state, cervical epithelium provides a structural and immunoprotective barrier mediated by the production of cytokines and antimicrobials [1]. Dilatation of the cervical canal is a component of many obstetric and gynecological procedures. It is required in gynecological procedures such as brachytherapy, preparation for operative hysteroscopy, and preparation for surgical abortion [2]. In obstetrics, cervical ripening is crucial for proper labor preinduction. Suitable labor preinduction can prevent dysfunctional labor. Since the dawn of time, gynecological practitioners have sought to obtain the best method to achieve cervical dilatation through experimentation with various materials. The ideal dilatation agent should be clinically effective, easy to use, cost-effective, and safe for the patient. Over the years, many effective methods to dilatate the cervical canal have been developed [3]. The cervix is often prepared with osmotic or mechanical dilatators (both synthetic and organic) or with pharmacological methods. The latest studies show that no single cervical ripening method is superior, considering safety outcomes and efficiency [4]. Correct and appropriate cervical dilatation is pivotal for the proper and safe conduct of the required diagnostic and therapeutic procedures involving the cervix and the interior of the uterus [4]. In recent years, the role of osmotic dilatators (both synthetic and Laminaria) in gynecological and obstetric procedures has been expanding. Osmotic dilatators are medical implements used to dilatate and soften the cervix by swelling as they absorb fluid from the surrounding cervical tissue. Their composition differs depending on their origin. Therefore, we can distinguish natural or synthetic dilators.
The present study is an in-depth analysis of the available literature using the keywords: labor, induction of labor, cervical ripening, Laminaria, Dilapan, osmotic dilators. An analysis of the literature in the last 15 years was made, also taking into account older papers relating to the biology of the cervix. This article aims to review the mechanisms of action and applications of osmotic dilators for cervical ripening in the induction of labor and in gynecology procedures.
Anatomy of the Cervix
The human uterine cervix is a passive organ located at the lower end of the uterus. It is developed (together with the uterus and vagina) from Mullerian ducts. It can be divided into a supravaginal portion and a lower portion. The cervical canal runs between the internal and external os. The cervix consists of connective tissue, ground substance, and a small amount of smooth muscle that make up about 10% of its dry weight. In addition, fibroblasts and wandering cells can be found in the cervical tissue. The columnar epithelium lines the cervical canal. Its cells are responsible for the production of a large amount of mucus composed mainly of water, enzymes, glycoproteins, and ions. The extracellular matrix (ECM) contains a large amount of collagen (mainly type I and III), glycosaminoglycans (GAGs), and elastin fibers [5–7]. Collagen fibers are cross-linked together by covalent bonds, which provides collagen molecules stability. The ground substance of cervical tissue (cervical stroma) is formed of glycosaminoglycans (GAGs), which are responsible for maintaining cervical tissue hydration and strength. GAGs chains create covalent bonds with the protein core, leading to creation of proteoglycans. Proteoglycans subsequently create bonds with collagen fibers [6]. This close association plays a pivotal role in providing cervical strength. The composition of the cervical stroma is determined by fibroblasts, which synthesize and degrade the major extracellular matrix elements [8].
Cervical Ripening
In general, the process of cervical ripening is the result of an inflammatory process accompanied by enzymatic breakdown of the extracellular matrix. The whole process is under strict regulation by hormonal factors [6]. The main mediator of these changes are metalloproteinases (MMPs), which digest ECM components, leading to significant loosening of the previously tightly packed structure of the cervical stroma [9]. The main sources of MMPs are neutrophils and macrophages, and their concentration in the cervical tissue significantly increases at term [10,11]. The activity of these cells (and the activity of MMPs) is significantly stimulated by pro-inflammatory cytokines, nitric oxide, and prostaglandins [11–14]. The most important cytokines involved in this process are interleukin-1 (IL-1) and interleukin-8 (IL-8) [15,16]. IL-1 directly stimulates the activity of cyclooxygenase 2 (COX-2), which enhances the production of prostaglandins (PGs) and increases the activity of MMPs [14]. IL-8 is an extremely strong chemotactic factor for neutrophils, which increases their concentration in the cervical tissue [17]. Additionally, IL-8 greatly enhances secretion of nitric oxide (NO). Nitric oxide strongly induces local vasodilation (which intensifies inflow of inflammatory cells), increases the activity of pro-inflammatory cytokines, and stimulates PGs production in cervical stroma [6,18–21]. Hormones such as ACTH and CRH are extremely important factor regulating the whole process [22–24].
Overall, this process consists of a series of complex biochemical pathways that results in rearrangement of the cervical ECM. Subsequently, the cervix significantly relaxes and becomes more susceptible, which makes the cervix prone to dilation.
Osmotic Dilators
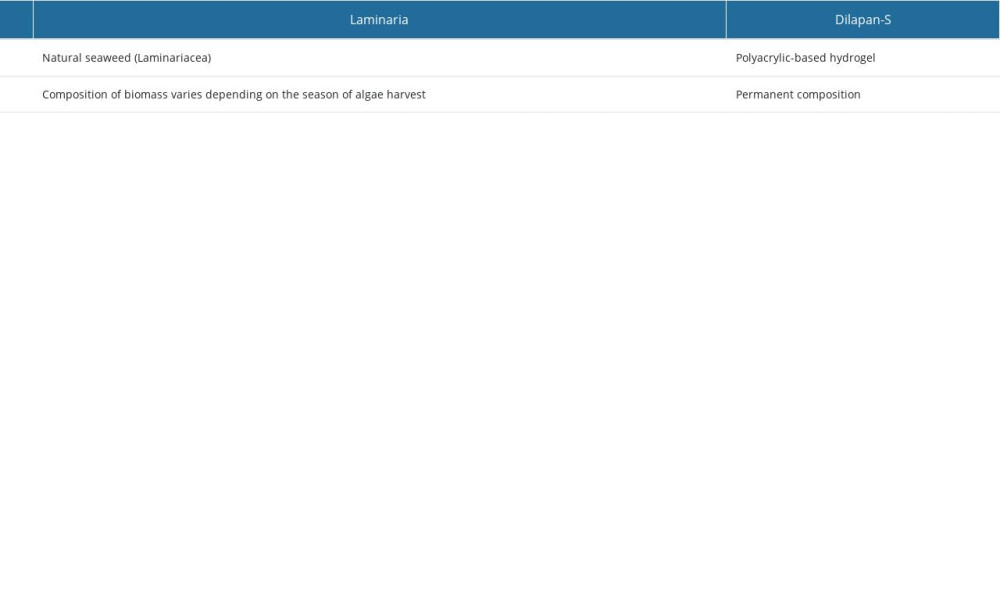
Osmotic dilatators are medical devices used to dilatate the cervix. They are dehydrated rods that can be placed in the cervical canal. The 2 types of osmotic dilatators currently available differ in origin: Laminaria and Dilapan-S. A Laminaria stick (of natural origin) is made from seaweed (
Although both types of osmotic dilators (Dilapan and Laminaria) are safe and highly effective, there are several dissimilarities between them [37], related mainly to the origins of Laminaria and their processing. They are created without modifications and result in properties depending on the portion of the plant from which the dilator is made [38]. Therefore, Laminaria has been reported to have several differences from synthetic osmotic dilators. Firstly, using Laminaria may be associated with an increased risk of infection because bacterial spores can remain in the tents despite sterilization of Laminaria [39,40]. Secondly, use of Laminaria can be associated with hypersensitivity reactions and anaphylaxis [41,42], believed to be mediated by IgE [43]. Finally, compared with Laminaria, synthetic dilators are entirely sterile, achieve larger cervical dilatation in a shorter timeframe, and swell more rapidly [44–47]. Additional advantages of synthetic dilators are the consistency of length and shape and, therefore, more predictable results (Table 1). The main disadvantage of the synthetic dilator is that Dilapan-S shortens as it swells (unlike Laminaria). Therefore, there is a theoretical possibility of dilator entrapment or fracture in the cervical canal [47]. Considering the above, we believe that synthetic dilators are more suitable and preferable, with fewer adverse effects in medical procedures involving cervical dilatation.
Mechanism of Action
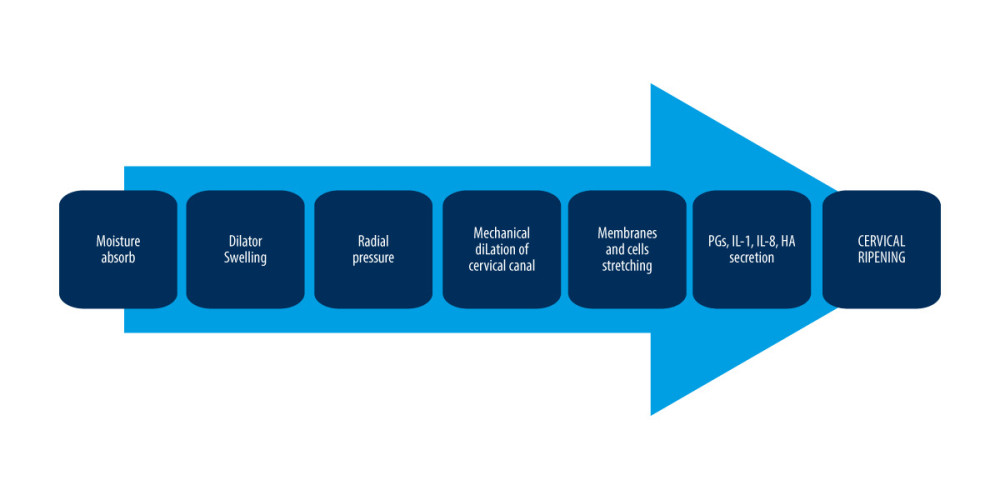
The mechanism of action of osmotic dilators has been known for years. However, recent studies clearly show that their mechanism of action is vastly more complex and multi-step than previously assumed. The basic principle of operation of osmotic dilators results from their hygroscopic nature. After application, these dilators absorb moisture (through their hygroscopic properties), drawing fluid from surrounding cervical tissue, which softens the tissue. As the dilators absorb fluid, axial expansion occurs, which applies a radial force to the walls of the cervical canal [37], subsequently leading to mechanical dilatation of the cervical canal. However, their impact on cervical tissue is not limited to a mechanical effect; ripening of the cervix is a process by which the cervix transforms from closed and rigid to soft and susceptible, and the cervical maturation process is complex, involving enzymatic breakdown, inflammatory reaction (accompanied by an infiltration of leukocytes), and endocrine regulation. Cervical remodeling is possible because of the reactions mediated by specific factors: nitric oxide, prostaglandins, and inflammatory cytokines. Osmotic dilators affect cervical tissue composition through the above factors [48]. Research has unequivocally shown that insertion of Laminaria in the cervical canal significantly increases the concentration of IL-1, IL-8, and PGE2, and elastase activity in cervical mucus [48–50]. IL-1 and IL-8 are basic factors involved in cervical ripening. IL-1 (occurring in 2 isoforms – IL-1α and IL-1β) can upregulate COX-2, which in turn increases the production of prostaglandins [51]. Additionally, IL-1 stimulates the production of other inflammatory cytokines involved in cervical ripening and enhances the expression of matrix metalloproteinases (MMPs), which can cleave collagen cross-links [1,6]. IL-8 is another important player in cervical ripening; it is a powerful neutrophil chemotactic agent that releases MMPs and inflammatory cytokines [6,52]. Moreover, IL-8 can increase vascular permeability, which subsequently increases the inflow of inflammatory cells [53,54]. IL-8 also stimulates fibroblasts to produce hyaluronic acid, which is another pivotal regulatory factor in cervical ripening [55,56]. Prostaglandins (PGs) are another important factor significantly participating in cervical maturation. PGs (mainly PGE2) can strongly stimulate (mainly via EP4 receptor) MMPs activity, upregulate IL-8 synthesis, and stimulate vasodilatation [47,56–58]. Apart from their impact on the cervical tissue, PGs also strongly affect uterus contractions. A possible explanation for the phenomenon of increased pro-inflammatory cytokines and PGs production during the use of Laminaria tents is that mechanical stretching of fetal membranes and myometrial cells (caused by Laminaria tent placement) dramatically increase secretion of factors such as PGs, IL-8, and hyaluronic acid (HA) [48,58–61] (Figure 1). Although the hygroscopic properties of osmotic dilators are considered the principal mechanism promoting cervical dilatation, osmotic dilators can drastically increase the production and release of PGs. Therefore, biochemical as well as mechanical factors may enhance cervical dilatation in women treated with osmotic dilators.
Brachytherapy
Brachytherapy is an extremely important part of oncological treatment, especially in gynecological oncology. It is a form of radiation therapy in which the radiation source is placed directly in or in close proximity to a malignant tumor [62]. Proper cervix preparation can significantly influence the brachytherapy procedure. Inadequate and forcible dilatation of the unprepared cervix can perforate the uterus or rupture the cervix, leading to life-threatening hemorrhage. Moreover, inadequate dilatation of the cervix before planned brachytherapy can result in incorrect placement of the applicator, which can significantly affect the effectiveness of the entire therapy [63]. Proper placement of the applicator is crucial for homogenous distribution of the radiation dose throughout the pelvis, ensuring adequate tumor coverage and sparing adjacent organs [64]. Theoretically, using osmotic dilators to dilate the cervical canal before planned brachytherapy can positively affect the correct placement of the applicator, resulting in the proper administration of radiation to diseased tissue. Research has shown that cervical osmotic dilation with a synthetic osmotic agent before planned brachytherapy can reduce treatment-associated morbidity and allow the delivery of adequate radiation therapy. Moreover, it can minimize discomfort associated with the procedure and thus eliminate the need for general or local anesthesia, which is highly important, especially in patients for whom anesthesia is contraindicated [65]. Finally, the absence of the need to use anesthesia significantly reduces the duration of the entire cervical brachytherapy procedure [65,66]. Additionally, placement of a Laminaria tent during pre-planning MRI for cervical cancer brachytherapy can improve the accuracy of image fusion with planning computed tomography (CT), which helps to delineate a more accurate brachytherapy profile [67]. Considering the above, we believe that using osmotic dilators prior to the cervical brachytherapy applicator placement can significantly improve the overall course of radiation therapy. However, this topic requires further studies.
Hysteroscopy
Hysteroscopy is a very important diagnostic and therapeutic tool in managing patients with infertility, suspected intrauterine cavity, and abnormal uterine bleeding [68]. It gained immense popularity due to its minimally invasive approach to intrauterine lesions. Adequate dilation of the cervical canal during operative hysteroscopy is one of the key elements of the entire procedure [69]. Cervical dilation can be challenging, particularly in nulligravidae, postmenopausal women, and women with cervical stenosis. Furthermore, some lengthy hysteroscopic operations (such as myomectomy) require proper cervical dilation to facilitate repeated insertions of the resectoscope. Inadequate cervical dilation prior to the hysteroscopy can lead to cervical laceration, uterine perforation, and the creation of a false passage during attempts to dilate the cervix [70]. Additionally, a proper degree of cervical dilation is required to extract previously excised lesions in the uterine cavity. In conclusion, proper cervical preparation is essential for conducting operative hysteroscopy and avoiding dangerous complications. Various agents have been tried to achieve cervical ripening of the cervix prior to operative hysteroscopy. These agents include osmotic dilators and prostaglandins (eg, misoprostol). However, it is still debatable which of the agents is superior in terms of cervical dilatation efficiency, safety of use, and possible adverse effects [71–74]. Recent studies clearly show that an osmotic dilator is more effective than misoprostol at achieving cervical ripening prior to operative hysteroscopy and has a lower percentage of adverse effects than vaginal misoprostol [73,75]. Moreover, an osmotic dilator achieved greater spontaneous dilation of the cervix than the vaginal misoprostol [74]. On the other hand, vaginal misoprostol seems to have an advantage due to its easy application, reduced cost, convenience, and patient acceptability [76]. However, using vaginal misoprostol in preoperative cervical ripening is associated with an increased rate of adverse effects such as febrile episodes, diarrhea, abdominal pain, and vaginal bleeding [75–78]. It has also been shown that intravaginal administration of misoprostol is more effective than oral administration in achieving preoperative cervical ripening [77]. Considering the above, we believe that osmotic dilators may be the superior tool to achieve cervical ripening prior to the operative hysteroscopy, with fewer adverse effects. Despite misoprostol’s advantages, it has a much higher percentage of adverse effects than osmotic dilators. It is advisable to explore this topic further to determine the appropriate dose of misoprostol with the fewest possible adverse effects.
Surgical Abortion
Induced surgical abortion is one of the most common surgeries performed worldwide. The vast majority of abortions are performed in the first trimester [79,80]. The entire procedure consists of curettage or evacuation preceded by dilatation of the cervical canal [79]. Proper cervical ripening before pregnancy termination by dilatation and suction curettage (D&C) greatly improves the procedure course and safety. Adequate cervical preparation is pivotal to safe abortion practice across all gestations. Forceful cervical dilatation is associated with cervical or uterine trauma with possible subsequent hemorrhage, infection, or adverse reproductive outcomes such as pregnancy loss, cervical insufficiency, and preterm birth [81]. Possible complications from D&C increase with advancing gestational age [81]. Therefore, the safety and success of the procedure largely depends on adequate cervical ripening. The cervix may be prepared with osmotic agents, pharmacological agents, or both. However, so far, it has not been possible to clearly establish which of the agents used to ripen the cervix has the best effectiveness and safety profile [82]. The most common pharmacological agent used to ripen the cervix prior to a surgical abortion is misoprostol. Its superiority lies in its effectiveness, low costs, and convenience of use with multiple routes of administration. The major disadvantage of misoprostol is its adverse effects, such as nausea, vomiting, and abdominal pain [82,83]. On the other hand, osmotic dilators are well-known cervical ripening agents with high efficiency and a small percentage of adverse effects. Their major drawbacks are the prolonged time required for cervical preparation and the need for vaginal examination with a speculum for proper application of the rod [84]. The main risks associated with using osmotic agents are pain, vasovagal reactions, allergic reactions, infection (rather when using Laminaria than synthetic dilators), and the possibility of dilator entrapment leading to intra-abdominal perforation [44,55,84,85–88]. Research shows that both Laminaria and misoprostol provide sufficient dilatation of the cervical canal for surgical abortion [87,88]. However, Laminaria seems to be a more effective cervical dilator (achieving greater dilatation of the cervix) than vaginal misoprostol for surgical abortion [87,90]. Moreover, using Laminaria is associated with a lower percentage of adverse effects such as nausea, vomiting, and vaginal bleeding [3,89–90]. Other studies comparing buccal misoprostol and osmotic dilators before surgical abortion have shown that either buccal misoprostol or osmotic dilator provides adequate preoperative dilatation of the cervical canal. However, the use of buccal misoprostol was associated with more pain [91]. Taking the above into account, we believe that although misoprostol has a similar effectiveness and a much easier route of administration, osmotic dilators appear to be slightly more effective due to the greater dilatation of the cervical canal and have a lower percentage of adverse effects in the maturation of the cervix before surgical abortion. Also noteworthy is the comparison of the effectiveness of natural (Laminaria) and synthetic (Dilapan-S) osmotic dilators. Research has unequivocally shown that Dilapan-S is superior to Laminaria in cervical ripening prior to surgical abortion. Moreover, the use of synthetic dilators allows greater dilatation of the cervix in a shorter time [44,45,88,89]. A puzzling issue is the simultaneous use of Laminaria and misoprostol for the induction of abortion. Interestingly, concurrent insertion of Laminaria and vaginal misoprostol did not improve the abortifacient effect of misoprostol [89]. Moreover, the addition of misoprostol to osmotic dilators was not found to increase cervical dilation [90]. Considering all the above, we believe that cervical preparation with osmotic dilators or misoprostol before surgical abortion is safe and effective. However, we suggest that osmotic dilators may be superior to misoprostol for cervical dilation and have fewer adverse effects.
Labor Preinduction
Over 20% of pregnant women undergo labor induction, and approximately half of them require a ripening agent due to an unfavorable cervix [92,93]. Cervical ripening is a process involving physically detectable softening, shortening, and dilatation of the uterine cervix [6]. The ripening process is a multifactorial process governed by endocrine changes, inflammatory response (accompanied by infiltration of leukocytes), and other biological changes. Research is ongoing to better understand this process’s molecular and biochemical basis [94]. Currently, prostaglandins, progesterone antagonists, and mechanical devices (eg, Foley catheter) have been used to ripen the cervix. Each of the above methods has a different degree of effectiveness and safety. No single cervical ripening method seems to be superior in safety and efficacy [4]. Prostaglandins are highly effective in cervical ripening. However, using prostaglandins (mainly misoprostol) is associated with significant adverse effects such as uterine hyperstimulation, uterine rupture (especially in previous cesarean sections), fetal heart rate changes, and fetal hypoxia. Additionally, use of prostaglandins is associated with nausea, vomiting, and abdominal pain [95]. Inflatable balloon catheters are also known to be strongly effective in cervical ripening, with fewer adverse effects, but they can cause cervical tissue injury and scarring. Moreover, the application of balloon catheters (eg, Foley catheter) can be associated with an increased risk of umbilical cord prolapse [96–98]. The role of osmotic dilators (both synthetic and natural) in labor preinduction has been constantly growing. Recent studies show that using osmotic dilators in labor preinduction is highly effective, with a small number of adverse effects and with minimal potential to cause uterine hyperstimulation [99–103]. As mentioned earlier, there are significant differences between Laminaria and synthetic osmotic dilators, with a possible higher rate of adverse effects when using Laminaria. In labor preinduction, the use of synthetic osmotic dilators (eg, Dilapan-S) appears to be preferable because it can shorten the induction-to-delivery interval, with fewer devices required to obtain significant cervical dilatation and with fewer adverse effects [104]. Patient satisfaction is an important factor determining the effectiveness of a cervical ripening agent. Compared to the Foley balloon, patients were more satisfied with a synthetic osmotic dilator in terms of time to relaxation and performance of daily activities [98]. Osmotic dilators are extremely effective in preinduction of labor after cesarean section. Additionally, their use reduces the risk of uterine hyperstimulation and rupture, which is of great importance in the preinduction of labor after cesarean sections [105–107]. Combining a synthetic osmotic dilator with vaginal prostaglandins or a progesterone antagonist (mifepristone) may lead to more effective cervical ripening and more rapid induction-to-delivery interval than prostaglandins or mifepristone alone [108]. However, the benefits of the combined use of vaginal prostaglandins and synthetic osmotic dilators are outweighed by an increased incidence of intrauterine infections [108].
Considering all the above, we believe that osmotic dilators are highly effective in labor preinduction. Thanks to their properties, they have a low percentage of adverse effects and are well tolerated by patients, which is a pivotal factor in the proper preinduction of labor.
Future Directions
This article aims to review the mechanisms of action and applications of osmotic dilators for cervical ripening in the induction of labor and in gynecology procedures.
The main emphasis was placed on the role of osmotic dilators in specific clinical situations (both gynecological and obstetrical), detailing the safety profile and efficiency of using dilators. We limited the information on the anatomy and molecular biology of the cervix to the minimum necessary to understand the mechanism of action of osmotic dilators. The data presented above indicate that osmotic dilators are extremely effective in preparing the cervix for planned diagnostic and therapeutic procedures. We suggest that their effectiveness is mainly due to their dual action. Firstly, due to the ability to absorb water from surrounding tissues (thanks to their hygroscopic properties), they can apply a radial force to the cervical canal, leading to cervical dilatation. Secondly, mechanical stretching of myometrial cells and fetal membranes can increase the secretion of PGs and IL-8, which are strong cervical ripening factors.
Natural osmotic dilators (Laminaria) are a puzzling issue that requires special attention. Although they are less effective and have a greater number of adverse effects compared to synthetic dilators, they may be useful in other situations. Taking all the above information into account, we believe that using synthetic osmotic dilators (in comparison with natural osmotic dilators) is more effective and much safer for the patient, with fewer adverse effects. Considering surgical abortion, the available data unequivocally suggest that osmotic dilators are more effective cervical dilators than are pharmacological agents. However, using osmotic dilators may also be associated with a small spectrum of adverse effects (as are other cervical ripening agents), the possibility of dilator entrapment, and the prolonged time required to achieve proper cervical dilatation. Further research is necessary to accurately assess the risk of adverse effects and impact on quality of life regarding cervical preparation and the patient’s preferences for using osmotic dilators and pharmacological agents. It is also worth noting that when comparing synthetic and natural osmotic dilators, it is possible to achieve greater cervical dilatation with a much shorter time using synthetic dilators. Therefore, we believe that the use of synthetic osmotic dilators prior to a surgical abortion is not only more effective but also has fewer adverse effects. The concurrent insertion of Laminaria and vaginal misoprostol did not improve the abortifacient effect of misoprostol. However, no data are available on the simultaneous use of a synthetic osmotic dilator with a pharmacological agent (eg, misoprostol). Bearing in mind the effectiveness of using a synthetic dilator, further research should evaluate the effect of the concurrent use of misoprostol with a synthetic osmotic dilator (Dilapan-S).
An extremely interesting topic is the use of osmotic dilators in the preinduction of labor. Presented data clearly indicate that osmotic dilators (in particular synthetic dilators) are highly effective in cervical ripening, with a small number of adverse effects (especially with minimal potential to cause uterine hyperstimulation). Moreover, the simultaneous application of a synthetic osmotic dilator with vaginal prostaglandins (or progesterone antagonist) may lead to much more effective cervical ripening.
Detailed knowledge of the mechanisms responsible for the maturation of the cervix is pivotal for the safe and proper preparation of the cervix. Therefore, understanding the events that lead to cervical ripening can lead to the development of more effective and safer labor induction methods. Moreover, comprehensive knowledge of cervical biology and selecting the appropriate cervical ripening agent can significantly influence delivery success and may prevent women from having dysfunctional labor. Therefore, we believe that it is necessary to further expand the (already extensive) knowledge in the field of cervical biology.
We also believe that synthetic osmotic dilators are a highly effective form of cervical ripening agents, as proven by many studies. Moreover, it is important to realize that brown algae (a natural dilator) are not constant in their biomass composition. The harvesting season of brown algae significantly affects their composition, which may affect their effectiveness and possibly cause adverse effects. However, further clinical trials and studies are required to more accurately assess the effect of changes in brown algae biomass on their effectiveness. As mentioned, Laminaria is composed mainly of cellulose, laminarin, and ions. Laminarin is a mucopolysaccharide that has versatile biomedical potential. Recent studies show that laminarin has antioxidant properties and can be used in cancer therapy or tissue engineering [109]. However, this requires more careful research to clearly define the effects of this substance on the tissues of the female reproductive system. Moreover, the use of Laminaria probably contributes to some extent to the detachment of placenta [110]. We believe that the substances contained in Laminaria can influence the fibrinolytic activity. However, this topic requires further, in-depth research to determine the exact effect of Laminaria on hemostasis and their possible applications.
Conclusions
According to the presented data, osmotic dilators are effective cervical ripening agents and therefore can be effectively used in gynecological and obstetric procedures. They can be successfully used in the induction of labor, preparation for surgical abortion, or as a preparation for elective brachytherapy in the case of endometrial cancer or cervical cancer.
We suggest that more research is needed to carefully compare the efficacy, possible adverse effects, and patient satisfaction of using the most commonly used cervical maturation (such as pharmacological agents or Foley catheter) with osmotic dilators. Careful study of this topic will allow selection of the most appropriate cervical ripening agent (with the best possible safety profile) and may significantly influence the success of delivery and prevent women from experiencing dysfunctional labor.
References
1. Leppert PC, Anatomy and physiology of cervical ripening: Clin Obstet Gynecol, 1995; 38(2); 267-79
2. Tenore JL, Methods for cervical ripening and induction of labor: Am Fam Physician, 2003; 67(10); 2123-28
3. Newmann SJ, Dalve-Endres A, Diedrich JT, Cervical preparation for second trimester dilation and evacuation: Cochrane Database Syst Rev, 2010(8); CD007310
4. Blanc-Petitjean P, Carbonne B, Deneux-Tharaux CMEDIP study group, Comparison of effectiveness and safety of cervical ripening methods for induction of labour: A population-based study using coarsened exact matching: Paediatr Perinat Epidemiol, 2019; 33(5); 313-22
5. Singer A, Jordan JA, The functional anatomy of the cervix, the cervical epithelium and the stroma: The Cervix, 2009; 13-37
6. Nicoll AE: The physiology of cervical ripening and the induction of labour: a potential role for the nitric oxide donor isosorbide mononitrate, 2001
7. Uldbjerg N, Ekman G, Malmström A, Ripening of the human uterine cervix related to changes in collagen, glycosaminoglycans, and collagenolytic activity: Am J Obstet Gynecol, 1983; 147(6); 662-66
8. Akgul Y, Holt R, Mummert M, Word A, Mahendroo M, Dynamic changes in cervical glycosaminoglycan composition during normal pregnancy and preterm birth: Endocrinology, 2012; 153(7); 3493-503
9. Osmers R, Rath W, Adelmann-Grill BC, Origin of cervical collagenase during parturition: Am J Obstet Gynecol, 1992; 166(5); 1455-60
10. Nagase H, Woessner JF, Matrix metalloproteinases: J Biol Chem, 1999; 274(31); 21491-94
11. Ledingham MA, Denison FC, Riley SC, Norman JE, Matrix metalloproteinases-2 and -9 and their inhibitors are produced by the human uterine cervix but their secretion is not regulated by nitric oxide donors: Hum Reprod, 1999; 14(8); 2089-96
12. Sennström MB, Brauner A, Byström B, Matrix metalloproteinase-8 correlates with the cervical ripening process in humans: Acta Obstet Gynecol Scand, 2003; 82(10); 904-11
13. Bollapragada S, Youssef R, Jordan F, Term labor is associated with a core inflammatory response in human fetal membranes, myometrium, and cervix: Am J Obstet Gynecol, 2009; 200(1); 104.e1-11 Erratum in: Am J Obstet Gynecol. 2009;201(2): 214
14. Sennström MB, Ekman G, Westergren-Thorsson G, Human cervical ripening, an inflammatory process mediated by cytokines: Mol Hum Reprod, 2000; 6(4); 375-81
15. Sykes L, MacIntyre DA, Teoh TG, Bennett PR, Anti-inflammatory prostaglandins for the prevention of preterm labour: Reproduction, 2014; 148(2); R29-40
16. el Maradny E, Kanayama N, Halim A, The effect of interleukin-1 in rabbit cervical ripening: Eur J Obstet Gynecol Reprod Biol, 1995; 60(1); 75-80
17. Winkler M, Fischer DC, Hlubek M, Interleukin-1beta and interleukin-8 concentrations in the lower uterine segment during parturition at term: Obstet Gynecol, 1998; 91(6); 945-49
18. Väisänen-Tommiska MR, Nitric oxide in the human uterine cervix: Endogenous ripening factor: Ann Med, 2008; 40(1); 45-55
19. Salvemini D, Masferrer JL, Interactions of nitric oxide with cyclooxygenase: In vitro, ex vivo, and in vivo studies: Methods Enzymol, 1996; 269; 12-25
20. Ekerhovd E, Weijdegård B, Brännström M, Nitric oxide induced cervical ripening in the human: Involvement of cyclic guanosine monophosphate, prostaglandin F(2 alpha), and prostaglandin E(2): Am J Obstet Gynecol, 2002; 186(4); 745-50
21. Väisänen-Tommiska M, Mikkola TS, Ylikorkala O, Misoprostol induces cervical nitric oxide release in pregnant, but not in nonpregnant, women: Am J Obstet Gynecol, 2005; 193(3 Pt 1); 790-96
22. Klimaviciute A, Calciolari J, Bertucci E, Corticotropin-releasing hormone, its binding protein and receptors in human cervical tissue at preterm and term labor in comparison to non-pregnant state: Reprod Biol Endocrinol, 2006; 4; 29
23. Li W, Challis JR, Corticotropin-releasing hormone and urocortin induce secretion of matrix metalloproteinase-9 (MMP-9) without change in tissue inhibitors of MMP-1 by cultured cells from human placenta and fetal membranes: J Clin Endocrinol Metab, 2005; 90(12); 6569-74
24. Aggelidou E, Hillhouse EW, Grammatopoulos DK, Up-regulation of nitric oxide synthase and modulation of the guanylate cyclase activity by corticotropin-releasing hormone but not urocortin II or urocortin III in cultured human pregnant myometrial cells: Proc Natl Acad Sci USA Mar 5, 2002; 99(5); 3300-5
25. Rubin B, Laminaria digitata: A checkered career: Econ Bot, 1977; 31(1); 66-71
26. Yoneshigue-Valentin Y: Hydrobiologia, 1990; 204; 461-66
27. Horn SJ, Moen E, Østgaard K, Direct determination of alginate content in brown algae by near infra-red (NIR) spectroscopy: J Appl Phycol, 1999; 11; 9-13
28. Black W, The seasonal variation in weight and chemical composition of the common British Laminariaceae: J Mar Biol Assoc UK, 1950; 29(1); 45-72
29. Black WAP, The seasonal variation in chemical constitution of some of the sub-littoral seaweeds common to scotland. Part III. Laminaria saccharina and saccorhiza bulbosa: Journal of the Society of Chemical Industry, 1948; 67; 172-76
30. Nelson TE, Lewis BA, Separation and characterization of the soluble and insoluble components of insoluble laminaran: Carbohydr Res, 1974; 33(1); 63-74
31. Adams JM, Ross AB, Anastasakis K, Seasonal variation in the chemical composition of the bioenergy feedstock Laminaria digitata for thermochemical conversion: Bioresour Technol, 2011; 102(1); 226-34
32. Ito K, Hori K, Seaweed: Chemical composition and potential food uses: Food Reviews International, 2009; 5; 101-44
33. Schiener P, Black KD, Stanley MS: J Appl Phycol, 2015; 27; 363-73
34. Rosell K-G, Srivastava LM, Seasonal variation in the chemical constituents of the brown algae Macrocystis integrifolia and Nereocystis luetkeana: Canadian Journal of Botany, 2011; 62(11); 2229-36
35. Fleurence J, Seaweed proteins: Biochemical, nutritional aspects and potential uses: Trends Food Sci Technol, 1999; 10; 25-28
36. Adams JMM, Ross AB, Aanstasakis K, Seasonal variation in the chemical composition of the bioenergy feedstock Laminaria digitata for thermochemical conversion: Bioresour Technol, 2011; 102; 226-34
37. Drunecký T, Reidingerová M, Plisová M, Experimental comparison of properties of natural and synthetic osmotic dilators: Arch Gynecol Obstet, 2015; 292(2); 349-54
38. Lichtenberg ES, Complications of osmotic dilators: Obstet Gynecol Surv, 2004; 59(7); 528-36
39. Sutkin G, Capelle SD, Schlievert PM, Creinin MD, Toxic shock syndrome after laminaria insertion: Obstet Gynecol, 2001; 98(5 Pt 2); 959-61
40. Acharya PS, Gluckman SJ, Bacteremia following placement of intracervical laminaria tents: Clin Infect Dis, 1999; 29(3); 695-97
41. Nguyen MT, Hoffman DR, Anaphylaxis to laminaria: J Allergy Clin Immunol, 1995; 95(1 Pt 1); 138-39
42. Chanda M, Mackenzie P, Day JH, Hypersensitivity reactions following laminaria placement: Contraception, 2000; 62(2); 105-6
43. Kim SH, Chang YH, Kim WK, Two cases of anaphylaxis after laminaria insertion: J Korean Med Sci, 2003; 18(6); 886-88
44. Allen RH, Goldberg ABBoard of Society of Family Planning, Cervical dilation before first-trimester surgical abortion (<14 weeks’ gestation). SFP Guideline 20071: Contraception, 2007; 76(2); 139-56
45. Fox MC, Krajewski CM, Cervical preparation for second-trimester surgical abortion prior to 20 weeks’ gestation: SFP Guideline #2013-4: Contraception, 2014; 89(2); 75-84
46. Bokström H, Wiqvist N, Prostaglandin release from human cervical tissue in the first trimester of pregnancy after preoperative dilatation with hygroscopic tents: Prostaglandins, 1995; 50(4); 179-88
47. Ye BL, Yamamoto K, Tyson JE, Functional and biochemical aspects of laminaria use in first-trimester pregnancy termination: Am J Obstet Gynecol, 1982; 142(1); 36-39
48. El Maradny E, Kanayama N, Halim A, Biochemical changes in the cervical mucus after application of laminaria tent: Acta Obstet Gynecol Scand, 1996; 75(3); 203-7
49. Kniss DA, Zimmerman PD, Garver CL, Fertel RH, Interleukin-1 receptor antagonist blocks interleukin-1-induced expression of cyclooxygenase-2 in endometrium: Am J Obstet Gynecol, 1997; 177(3); 559-67
50. Ogawa M, Hirano H, Tsubaki H, The role of cytokines in cervical ripening: Correlations between the concentrations of cytokines and hyaluronic acid in cervical mucus and the induction of hyaluronic acid production by inflammatory cytokines by human cervical fibroblasts: Am J Obstet Gynecol, 1998; 179(1); 105-10
51. Watari M, Watari H, DiSanto ME, Pro-inflammatory cytokines induce expression of matrix-metabolizing enzymes in human cervical smooth muscle cells: Am J Pathol, 1999; 154(6); 1755-62
52. Winkler M, Fischer DC, Ruck P, Parturition at term: Parallel increases in interleukin-8 and proteinase concentrations and neutrophil count in the lower uterine segment: Hum Reprod, 1999; 14(4); 1096-100
53. Lindsey JD, Kashiwagi K, Boyle D, Prostaglandins increase proMMP-1 and proMMP-3 secretion by human ciliary smooth muscle cells: Curr Eye Res, 1996; 15(8); 869-75
54. Denison FC, Calder AA, Kelly RW, The action of prostaglandin E2 on the human cervix: Stimulation of interleukin 8 and inhibition of secretory leukocyte protease inhibitor: Am J Obstet Gynecol, 1999; 180(3 Pt 1); 614-20
55. Kloeck FK, Jung H, In vitro release of prostaglandins from the human myometrium under the influence of stretching: Am J Obstet Gynecol, 1973; 115(8); 1066-69
56. Maradny EE, Kanayama N, Halim A, Stretching of fetal membranes increases the concentration of interleukin-8 and collagenase activity: Am J Obstet Gynecol, 1996; 174(3); 843-49
57. Kanayama N, Fukamizu H, Mechanical stretching increases prostaglandin E2 in cultured human amnion cells: Gynecol Obstet Invest, 1989; 28(3); 123-26
58. Takemura M, Itoh H, Sagawa N, Cyclic mechanical stretch augments hyaluronan production in cultured human uterine cervical fibroblast cells: Mol Hum Reprod, 2005; 11(9); 659-65
59. El Maradny E, Kanayama N, Kobayashi H, The role of hyaluronic acid as a mediator and regulator of cervical ripening: Hum Reprod, 1997; 12(5); 1080-88
60. Norman M, Ekman G, Malmström A, Changed proteoglycan metabolism in human cervix immediately after spontaneous vaginal delivery: Obstet Gynecol, 1993; 81(2); 217-23
61. Christiaens I, Zaragoza DB, Guilbert L, Inflammatory processes in preterm and term parturition: J Reprod Immunol, 2008; 79(1); 50-57
62. Harkenrider MM, Kamrava M, Embracing a future of progress in gynecologic brachytherapy: Brachytherapy, 2023; 22(1); 6-8
63. Corn BW, Hanlon AL, Pajak TF, Technically accurate intracavitary insertions improve pelvic control and survival among patients with locally advanced carcinoma of the uterine cervix: Gynecol Oncol, 1994; 53(3); 294-300
64. Viswanathan AN, Moughan J, Small W, The quality of cervical cancer brachytherapy implantation and the impact on local recurrence and disease-free survival in radiation therapy oncology group prospective trials 0116 and 0128: Int J Gynecol Cancer, 2012; 22(1); 123-31
65. Mayr NA, Sorosky JI, Zhen W, The use of laminarias for osmotic dilation of the cervix in gynecological brachytherapy applications: Int J Radiat Oncol Biol Phys, 1998; 42(5); 1049-53
66. Glynn KM, Riker AI, Anesthetic considerations for intraoperative radiation therapy: Ochsner J, 2015; 15(4); 438-40
67. Takagawa Y, Izumi S, Kita M, Laminaria tent insertion in preplanning MRI for CT-based cervical cancer brachytherapy: Brachytherapy, 2022; 21(2); 170-76
68. Marchand GJ, Masoud AT, Ulibarri H, Effect of the decision to perform hysteroscopy on asymptomatic patients before undergoing assisted reproduction technologies – a systematic review and meta-analysis: AJOG Glob Rep, 2023; 3(2); 100178
69. Chiofalo B, Calandra M, Bruno V, Outpatient hysteroscopic polypectomy – a retrospective study comparing rigid and semirigid office hysteroscopes: Diagnostics (Basel), 2023; 13(5); 988
70. Bradley LD, Complications in hysteroscopy: Prevention, treatment and legal risk: Curr Opin Obstet Gynecol, 2002; 14(4); 409-15
71. Al-Fozan H, Firwana B, Al Kadri H, Preoperative ripening of the cervix before operative hysteroscopy: Cochrane Database Syst Rev, 2015(4); CD005998
72. Arena S, Zupi E, Affronti G, Cervical ripening prior to hysteroscopy: Is the application of misoprostol useful?: Minerva Ginecol, 2011; 63(5); 439-48
73. Yu D, Li TC, Xia E, A prospective, randomized, controlled trial comparing vaginal misoprostol and osmotic dilator in achieving cervical ripening before operative hysteroscopy: Gynecol Surg, 2006; 3; 186-89
74. Karakus S, Akkar OB, Yildiz C, Comparison of effectiveness of laminaria versus vaginal misoprostol for cervical preparation before operative hysteroscopy in women of reproductive age: A prospective randomized trial: J Minim Invasive Gynecol, 2016; 23(1); 46-52
75. Darwish AM, Ahmad AM, Mohammad AM, Cervical priming prior to operative hysteroscopy: A randomized comparison of laminaria versus misoprostol: Hum Reprod, 2004; 19(10); 2391-94
76. Crane JM, Healey S, Use of misoprostol before hysteroscopy: A systematic review: J Obstet Gynaecol Can, 2006; 28(5); 373-79
77. da Costa AR, Pinto-Neto AM, Amorim M, Use of misoprostol prior to hysteroscopy in postmenopausal women: A randomized, placebo-controlled clinical trial: J Minim Invasive Gynecol, 2008; 15(1); 67-73
78. Batukan C, Ozgun MT, Ozcelik B, Cervical ripening before operative hysteroscopy in premenopausal women: A randomized, double-blind, placebo-controlled comparison of vaginal and oral misoprostol: Fertil Steril, 2008; 89(4); 966-73
79. Brittain JJ, Wahl SE, Strauss JF, Prior spontaneous or induced abortion is a risk factor for cervical dysfunction in pregnant women: A systematic review and meta-analysis: Reprod Sci, 2023 [Online ahead of print]
80. Finer LB, Henshaw SK, Abortion incidence and services in the United States in 2000: Perspect Sex Reprod Health, 2003; 35(1); 6-15
81. Zhou W, Olsen J, Are complications after an induced abortion associated with reproductive failures in a subsequent pregnancy?: Acta Obstet Gynecol Scand, 2003; 82(2); 177-81
82. : Clinical Guidelines Cervical dilation before first-trimester surgical abortion (b14 weeks’ gestation), 2007
83. Lauersen NH, Den T, Iliescu C, Cervical priming prior to dilatation and evacuation: A comparison of methods: Am J Obstet Gynecol, 1982; 144(8); 890-94
84. Harjee R, Bahrani R, Chan M, Osmotic dilator intra-abdominal perforation: A case report: J Obstet Gynaecol Can, 2020; 42(5); 637-39
85. Chambers DG, Willcourt RJ, Laver AR, Comparison of Dilapan-S and laminaria for cervical priming before surgical pregnancy termination at 17–22 weeks’ gestation: Int J Womens Health, 2011; 3; 347-52
86. Diedrich JT, Drey EA, Newmann SJ, Society of Family Planning clinical recommendations: Cervical preparation for dilation and evacuation at 20–24 weeks’ gestation: Contraception, 2020; 101(5); 286-92
87. Sagiv R, Mizrachi Y, Glickman H, Laminaria vs. vaginal misoprostol for cervical preparation before second-trimester surgical abortion: A randomized clinical trial: Contraception, 2015; 91(5); 406-11
88. Burnett MA, Corbett CA, Gertenstein RJ, A randomized trial of laminaria tents versus vaginal misoprostol for cervical ripening in first trimester surgical abortion: J Obstet Gynaecol Can, 2005; 27(1); 38-42
89. Jain JK, Mishell DR, A comparison of misoprostol with and without laminaria tents for induction of second-trimester abortion: Am J Obstet Gynecol, 1996; 175(1); 173-77
90. Chodankar R, Gupta J, Gdovinova D, Synthetic osmotic dilators for cervical preparation prior to abortion – an international multicentre observational study: Eur J Obstet Gynecol Reprod Biol, 2018; 228; 249-54
91. Bartz D, Maurer R, Allen RH, Buccal misoprostol compared with synthetic osmotic cervical dilator before surgical abortion: A randomized controlled trial: Obstet Gynecol, 2013; 122(1); 57-63
92. Sium AF, Prager S, Wolderufael M, Foley catheter for cervical preparation prior to second trimester dilation and evacuation: A supply-based alternative for surgical abortion: A case series: Contracept X, 2022; 4; 100085
93. Nicholson JM, Kellar LC, Henning GF, The association between the regular use of preventive labour induction and improved term birth outcomes: Findings of a systematic review and meta-analysis: BJOG, 2015; 122(6); 773-84
94. Ducarme G, Martin S, Chesnoy V, Prospective observational study investigating the effectiveness, safety, women’s experiences and quality of life at 3 months regarding cervical ripening methods for induction of labor at term-The MATUCOL study protocol: PLoS One, 2022; 17(1); e0262292
95. Witter FR, Prostaglandin E2 preparations for preinduction cervical ripening: Clin Obstet Gynecol, 2000; 43(3); 469-74
96. Hasegawa J, Sekizawa A, Ikeda T, The use of balloons for uterine cervical ripening is associated with an increased risk of umbilical cord prolapse: population based questionnaire survey in Japan: BMC Pregnancy Childbirth, 2015; 15; 4
97. Gommers JSM, Diederen M, Wilkinson C, Risk of maternal, fetal and neonatal complications associated with the use of the transcervical balloon catheter in induction of labour: A systematic review: Eur J Obstet Gynecol Reprod Biol, 2017; 218; 73-84
98. Shindo R, Aoki S, Yonemoto N, Hygroscopic dilators vs balloon catheter ripening of the cervix for induction of labor in nulliparous women at term: Retrospective study: PLoS One, 2017; 12(12); e0189665
99. Gupta J, Chodankar R, Baev O, Synthetic osmotic dilators in the induction of labour – an international multicentre observational study: Eur J Obstet Gynecol Reprod Biol, 2018; 229; 70-75
100. Blumenthal PD, Ramanauskas R, Randomized trial of Dilapan and Laminaria as cervical ripening agents before induction of labor: Obstet Gynecol, 1990; 75(3 Pt 1); 365-68
101. Lackritz R, Gibson M, Frigoletto FD, Preinduction use of laminaria for the unripe cervix: Am J Obstet Gynecol, 1979; 134(3); 349-50
102. Crosby DA, O’Reilly C, McHale H, A prospective pilot study of Dilapan-S compared with Propess for induction of labour at 41+ weeks in nulliparous pregnancy: Ir J Med Sci, 2018; 187(3); 693-99
103. Kazzi GM, Bottoms SF, Rosen MG: Obstet Gynecol, 1982; 60(4); 440-43
104. Saad AF, Villarreal J, Eid J, A randomized controlled trial of Dilapan-S vs Foley balloon for preinduction cervical ripening (DILAFOL trial): Am J Obstet Gynecol, 2019; 220(3); 275e1-e9
105. Maier JT, Metz M, Watermann N, Induction of labor in patients with an unfavorable cervix after a cesarean using an osmotic dilator versus vaginal prostaglandin: J Perinat Med, 2018; 46(3); 299-307
106. Koenigbauer JT, Schalinski E, Jarchau U, Cervical ripening after cesarean section: a prospective dual center study comparing a mechanical osmotic dilator vs. prostaglandin E2: J Perinat Med, 2021; 49(7); 797-805
107. Hibbard JU, Shashoua A, Adamczyk C, Ismail M, Cervical ripening with prostaglandin gel and hygroscopic dilators: Infect Dis Obstet Gynecol, 1998; 6(1); 18-24
108. Baev OR, Babich DA, Prikhodko AM, A comparison between labor induction with only Dilapan-S and a combination of mifepristone and Dilapan-S in nulliparous women: A prospective pilot study: J Matern Fetal Neonatal Med, 2021; 34(17); 2832-37
109. Zargarzadeh M, Amaral AJR, Custódio CA, Mano JF, Biomedical applications of laminarin: Carbohydr Polym, 2020; 232; 115774
110. Jonasson A, Larsson B, Lecander I, Astedt B, Placental and decidual u-PA, t-PA, PAI-1 and PAI-2 concentrations, as affected by cervical dilatation with laminaria tents or Hegar dilators: Thromb Res, 1989; 53(2); 91-97
In Press
07 May 2024 : Clinical Research
Treatment of AVN-Induced Proximal Pole Scaphoid Nonunion Using a Fifth and Fourth Extensor Compartmental Ar...Med Sci Monit In Press; DOI: 10.12659/MSM.944553
16 Mar 2024 : Clinical Research
Diagnostic Efficiency of ACR-TIRADS Score for Differentiating Benign and Malignant Thyroid Nodules of Vario...Med Sci Monit In Press; DOI: 10.12659/MSM.943228
08 May 2024 : Clinical Research
Effect of Individualized PEEP Guided by Driving Pressure on Diaphragm Function in Patients Undergoing Lapar...Med Sci Monit In Press; DOI: 10.12659/MSM.944022
21 Mar 2024 : Clinical Research
Impact of Serum Vitamin D, B6, and B12 and Cognitive Functions on Quality of Life in Peri- and Postmenopaus...Med Sci Monit In Press; DOI: 10.12659/MSM.943249
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952